Luminescence
Luminescence. Debbie Ormond. Introduction. Luminescence is the emission of light by a substance without the need for heating. Examples of luminescence are fluorescence, phosphorescence and chemiluminescence . Uses of luminescence. Glow in the dark stickers. Fluorescent tube lighting.

Luminescence
E N D
Presentation Transcript
Luminescence Debbie Ormond
Introduction Luminescence is the emission of light by a substance without the need for heating. Examples of luminescence are fluorescence, phosphorescence and chemiluminescence.
Uses of luminescence Glow in the dark stickers Fluorescent tube lighting Glow sticks Forensics Biomarker (mouse brain) Glow in the dark loo roll
The Electromagnetic Spectrum Visible (420-700 nm) X-Rays Radio waves Microwaves Infra Red Ultra Violet Gamma Rays Wavelength (m) 1 102 10-2 10-4 10-6 10-8 10-12 10-10 Energy increases Wavelength increases The electromagnetic spectrum shows a continuous range of wavelengths. The shortest wavelengths belong to gamma rays and the longest belong to radio waves. The wavelengths we need to concentrate on for luminescence are the visible and ultra violet.
What is the name of the longest wavelength in the electromagnetic spectrum? Gamma rays Radio waves Visible light
What is the name of the largest energy in the electromagnetic spectrum? Gamma rays Infra red Ultraviolet
Electromagnetic spectrum Looking at the electromagnetic spectrum you can see that as the energy decreases, the wavelength increases. This is because of the relationship (h and c are constant values) h=Planck’s constant c=speed of light =wavelength
Luminescence Luminescence occurs when the energy in is ultra violet and the energy out is visible light. Luminescence occurs when an electron is excited by a photon (a packet) of light to a higher energy level. The electron then works its way down the energy levels before dropping back to the ground state and releasing a photon of light. The wavelength of the energy released corresponds to different areas of the electromagnetic spectrum.
What is the name for a packet of light? A proton A photon A parcel
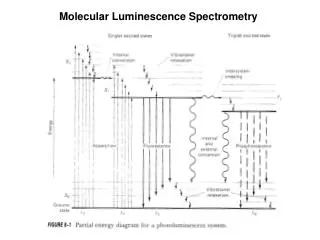
Think of luminescence like a walk up a mountain. You need a lot of energy to get to the top. However, on the way down you take a cable car for a bit and then stroll down the rest using a lot less energy. This is represented scientifically by the previous diagram (Jablonski diagram).
Below is a diagram representing what happens during luminescence. The diagram is called a Jablonski diagram. The circle represent the electron which is excited to a high energy level from the ground state. Is there a difference in energy between the Energy in and Energy out? Hint: Look at the length of the excite and relax lines Excitation Energy In has more energy Energy Out has more energy They have the same energy Relaxation Energy out (Emission) Energy in (Absorption) Ground state
If the Energy In was in the ultra violet region. What region would you expect the Energy Out to be in? Gamma rays Infra Red Visible light
Fluorescence and Phosphorescence Fluorescence and phosphorescence are both forms of luminescence. In fluorescence the electron falls down quickly releasing light for a short period. In phosphorescence the electron falls over a long period of time so the light is released for a longer period
The vials on the left and the rocks on the right all fluoresce under UV light. The bugs in the middle look like they are doing exactly the same, but when the lights go out the bugs are still glowing. This is phosphorescence. Click the video above
Key Concepts • Luminescence is when energy is absorbed and then emitted as visible light. • The energy emitted is less than the energy absorbed. • Fluorescence is when the light is emitted over a short period of time. • Phosphorescence is when the light is emitted over a long period of time.
THE END! Please close the presentation
Visible (420-700 nm) X-Rays Radio waves Microwaves Infra Red Ultra Violet Gamma Rays 1 102 10-2 10-4 10-5 10-8 10-12 10-10 Energy increases Wavelength increases Back
Well Done! Radio waves have the longest wavelength in the electromagnetic spectrum. Next question
Try re-answering the question bearing in mind that the longest wavelength is to the left of the electromagnetic spectrum. Electromagnetic spectrum Try again
Visible (420-700 nm) X-Rays Radio waves Microwaves Infra Red Ultra Violet Gamma Rays 1 102 10-2 10-4 10-5 10-8 10-12 10-10 Energy increases Wavelength increases Back
Well done! Gamma rays have the largest energy in the electromagnetic spectrum. Next
Have another go at the question keeping in mind that the largest energy is to the right of the electromagnetic spectrum. Electromagnetic spectrum Try again
Well done! A photon is the name for a packet of light. Next
Have a look back at the previous slide and have another go. Try again Previous slide
Well done! The energy in is larger than the energy out. Next
Have another look at the Jablonski diagram Hint: Imagine drawing a line from the ground state up to the top excitation level (energy in). Now draw one from the ground state to the bottom excitation level (energy out). Which one is longer? This is the one with more energy. Jablonski diagram
Visible (420-700 nm) X-Rays Radio waves Microwaves Infra Red Ultra Violet Gamma Rays 1 102 10-2 10-4 10-5 10-8 10-12 10-10 Energy increases Wavelength increases Back
Visible (420-700 nm) X-Rays Radio waves Microwaves Infra Red Ultra Violet Gamma Rays 1 102 10-2 10-4 10-5 10-8 10-12 10-10 Energy increases Wavelength increases Back
Try the question again but this time bear in mind that the difference in energy will not be that big and energy out is smaller than energy in. Try again Electromagnetic spectrum
Well done! In luminescence the energy in is usually in the ultra violet region and the energy out is visible light. This is how we can see things fluorescing. Next
Visible (420-700 nm) X-Rays Radio waves Microwaves Infra Red Ultra Violet Gamma Rays 1 102 10-2 10-4 10-5 10-8 10-12 10-10 Energy increases Wavelength increases Back