Understanding Stoichiometry and Chemical Reactions**
This guide delves into the essential concepts of stoichiometry and chemical equations. It explains how to balance chemical equations, the significance of coefficients in determining mole ratios, and the law of conservation of mass. The guide also covers practical problems involving mole-mole, mass-mole, and mass-mass conversions in chemical reactions. Additionally, it introduces the concept of limiting reactants, explaining how to identify them in reactions and their impact on product yield.

Understanding Stoichiometry and Chemical Reactions**
E N D
Presentation Transcript
EX: 2 Mg + O2 2 MgO reactants products Equation Stoichiometry Chemical Equation – indicates the reactants and productsin a rxn; it also tells you the relative amounts of reactantsand products involved in the rxn. Stoichiometry – the study of the mathematical relationshipsinvolving chemical formulas and equations.
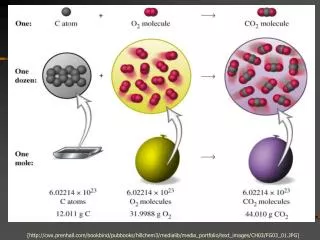
Chemical equations reflect the law of conservationof mass – matter is neither created nor destroyedin an ordinary chemical rxn. The mass of the products equals the massof the reactants that reacted. This is represented in an equation by having the same number of each atom on both sides of the equation. Coefficients are used to balance equations.
(Review) Coefficients can stand for: 1) The relative number of atoms, molecules, or formula units 2) The relative number of moles 3) The relative volume of gases EX: 2 H2 + O2 2 H2O 2 molecules H2 + 1 molecule O2 2 molecules H2O 2 mol H2 + 1 mol O2 2 mol H2O 2 volumes H2 + 1 volume O2 2 volumes H2O
What are the possible mole ratios for: 3 Mg + 2 P Mg3P2 ***Coefficients tell you the relative number of moles of each reactant and product involved in a rxn.*** mole ratio (coefficient ratio) – a conversion factor that allows you to convert from moles of one substance to moles of another substance in a rxn. EX: CH4 + 2 O2 CO2 + 2 H2O Example mole ratios:
Moles of given substance coefficient of substance sought coefficient of given substance = Moles of substance sought I.Mole – Mole Problems You are given moles of one substance in a rxn and must find the moles of another substance. Use the mole ratio from the equation to solve Mole-mole problems:
1. For the rxn N2 + 3 H2 2 NH3 a) How many moles of N2 will react with 8.0 mol of H2? 2. For the rxn C5H12O + 6 O2 5 CO2 + 3 H2O a) If 127 mol of CO2 are produced, how many moles of H2O are produced? Example mole-mole problems b) How many moles of NH3 can be made from 12.0 mol of H2? • If 15 mol of O2 react, how many moles of CO2 • are produced?
II. Mass-Mole, Mole-Mass, and Mass-Mass Problems A. Review: Molar Mass - used as a conversion factor to convert between grams (mass) of a substance and # of moles. 1) to convert grams to moles, use the following set up: 1 mol given grams X molar mass = moles 2) to convert moles to grams, use the following set up:molar mass given moles X 1 mol = grams
B. Mass-Mole Problem (2 steps) – you are given the mass(grams) of one substance in a rxn, and are asked to find the number of moles of another substance. EX: For the rxn N2 + 3 H2 2 NH3 1) How many moles of NH3 can you make from 28g of N2? 2) How many moles of H2 are needed to make 100. g of NH3?
C. Mole-Mass Problem (2 steps) – you are given the moles of one substance in a rxn, and are asked to find the number of grams (mass) of another substance. • EX: For the rxn CH4 + 2 O2 CO2 + 2 H2O • How many grams of H2O are produced by combusting2.25 x 1012 moles of CH4? • If 0.42 moles of O2 react, how many grams of CH4 react?
GIVEN GRAMS OF“A” (mass) GIVEN MOLES OF “A” MOLES OF “B”SOUGHT GRAMS OF “B” SOUGHT (mass) Suppose you have two substances, “A” and “B”, in a rxn. The following flow chart shows the stoichiometric relationship between them: 1 mol A . X molar mass of A molar mass X of B . 1 mol B coefficient of sought (B) X coefficient of given (A)
EX: For the rxn N2 + 3 H2 2 NH3 1) How many grams of H2 will react with 237g of N2? III. Mass-Mass Problems (3 steps) You are given the mass (grams) of one substance in a rxn, and are asked to find the number of grams (mass) of another substance. 2) How many grams of N2 do you need to produce 82.8 g NH3?
EX2: Decane, C10H22 , is a component of gasoline. 1) How many grams of CO2 will be produced by the complete combustion of 345g of C10H22 ? 2) What mass of oxygen reacted if 3.5 x 104 g of H2O was produced?
EX3: Aluminum metal can be produced by the electrolytic decomposition of aluminum oxide. • How many kilograms of Al can be produced by • decomposing one metric ton (1.0 x 103 kg) of aluminum oxide? 2) How many grams of oxygen would be consumed in the production of 3.5 x 106 g of Aluminum?
Cake Recipe To make one cake you need:3 eggs, 4 cups flour, 2 qts of milk, and 1 cup of sugar. You have: 12 eggs, 12 cups flour, 10 qts of milk, and 6 cups of sugar. How many cakes can you make?
In most reactions that occur, you don’t use “exact” amounts of each reactant. Usually one reactant gets used up before the other. limiting reactant – the reactant that gets totally used up in a rxn; it’s the reactant you run out of. (It “limits” the rxn.) excess reactant – any reactant that is not totally used up in a rxn; you still have some of it left after the rxn stops.
In this example, 10 molecules of H2 react with 7 molecules of O2, according to the equation: 2 H2 + O2 2 H2O The limiting reactant would be H2. The excess reactant would be O2.
Limiting Reactant Problems You are given the initial amounts of the reactants in a rxn, and must determine: a) which reactant is the limiting reactant. You also should be able to determine: b) how much product is produced, and c) how much of the excess reactant is left over. • **a) To determine which reactant is the limiting reactant:** • Find out how much product can be made from the 1st reactant • Find out how much product can be made from the 2nd reactant • Which ever reactant produces the LEAST product is the limiting reactant. (In other words: You need to do two mass-mass problems to figure out the limiting reactant.)
EX: 125g of Br2 and 75.0g Li are reacted together according to the equation: 2 Li + Br2 2 LiBr What is the limiting reactant?
**b) The amount of product actually produced is the amount produced by the limiting reactant.** In the example above, 136g of LiBr would actually be produced. EX: 24.0g of CH4 is burned in a closed container that holds 50.0g of O2. a) What is the limiting reactant? b) How many grams of CO2 is produced? • **c) To find out the amount of excess reactant left over:** • calculate the mass of excess reactant that reacted with the limiting reactant. (mass-mass problem) • subtract the amount of excess reactant that reacted (from step 1) from the original mass of excess reactant; this tells you how much was left over.
GIVEN GRAMS OF“A” (mass) GIVEN MOLES OF “A” MOLES OF “B”SOUGHT GRAMS OF “B” SOUGHT (mass) Suppose you have two substances, “A” and “B”, in a rxn. The following flow chart shows the stoichiometric relationship between them: 1 mol A . X molar mass of A molar mass X of B . 1 mol B coefficient of sought (B) X coefficient of given (A)
C6H12O6 + 6 O2 6 CO2 + 6 H2O How many grams of water could be made from 1.00 gram of glucose? Starting with 1.00 g of C6H12O6… we calculate the moles of C6H12O6… use the coefficients to find the moles of H2O… and then turn the moles of water to grams
Practice Limiting Reactant Problems EX: 50.0g of Na is reacted with 50.0g of Cl2 in synthesis reaction. a) what is the limiting reactant?b) how much NaCl is produced?c) what is the excess reactant, and how much of it remains after the rxn?
EX: N2H4 + 2 H2O2 N2 + 4 H2O 65.0g of N2H4 reacts with 75.0g of H2O2 a) what is the limiting reactant?b) how much N2 is produced?c) what is the excess reactant, and how much of it remains after the rxn?
Actual yield – the measured amount of product collected in an experiment. (The amount “actually” produced.) Theoretical and Percent Yield Theoretical Yield – the maximum amount of productthat can be produced in a rxn. Theoretical yield is determined by using the rxn’s equation and doing a mass-mass problem. (The amount that “theoretically”could have been produced.)
Actual Yield Theoretical Yield Percent Yield = x 100 Percent Yield A comparison of the amount actually obtained to the amount it was possible to make
Percent Yield Problems 1. In an experiment, 100. g of Na is reacted with excess Cl2. 239 g of NaCl is collected. a) What is the theoretical yield of NaCl? b) What is the percent yield of NaCl? 2. 150. g of O2 is reacted with excess Mg. 300. g of MgO is collected. a) What is the theoretical yield of MgO? b) What is the percent yield of MgO?
Solution Stoichiometry Problems
moles of solute Molarity (M) = liters of solution Review: Molarity Molarity is used to convert between volume of solutionand moles of dissolved solute. 1. How many moles of NaCl are in 3.5 L of 2.0 M NaCl? 2. How many moles AgNO3 are in 125 mL 3.00 M AgNO3?
VOLUME OF “A”GIVEN GIVEN GRAMS OF“A” (mass) GIVEN MOLES OF “A” MOLES OF “B”SOUGHT GRAMS OF “B” SOUGHT (mass) 1 mol A . X molar mass of A molar mass X of B . 1 mol B coefficient of sought (B) X coefficient of given (A)
Example Solution Stoich Problems 1) Excess Cu is placed in 250 mL of 0.50 M AgNO3 andthe following rxn occurs: 2 AgNO3 + Cu 2 Ag + Cu(NO3)2What mass of Ag can be produced? 2) 125 mL of 2.00 M Pb(NO3)2 is mixed with excess NaCl andthe following rxn occurs: 2 NaCl + Pb(NO3)2 PbCl2 + 2 NaNO3What mass of PbCl2 precipitates?