Periodic Table Review Activity
430 likes | 698 Views
Periodic Table Review Activity. Definition. What does PERIODIC mean? A repeating pattern Mendeleev left some spaces in his table because not all elements lined up according to atomic mass. He used info he knew about surrounding elements to PREDICT properties and masses of unknown elements.

Periodic Table Review Activity
E N D
Presentation Transcript
Definition • What does PERIODIC mean? • A repeating pattern • Mendeleev left some spaces in his table because not all elements lined up according to atomic mass. He used info he knew about surrounding elements to PREDICT properties and masses of unknown elements. How about that!
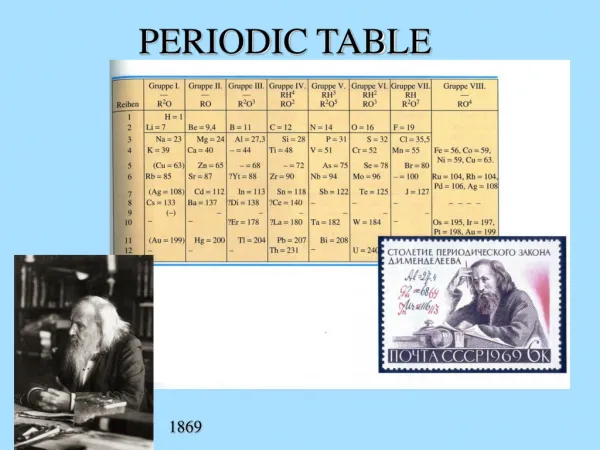
Mendeleev (1869) • Organized table by... • ATOMIC MASS • Thought pattern must exist between all elements • Elements in columns show similar properties… • Both physical and chemical! • Predicted properties of “missing” elements
Moseley (c. 1914) • rearranged table by ... • ATOMIC NUMBER • Determined atomic number of elements by determining number of protons in element
Seaborg (1944) • Rearranged Periodic Table with... • Lanthanide and Actinide Series pulled out/separated from main Periodic Table • Discovered countless isotopes & 10 new elements
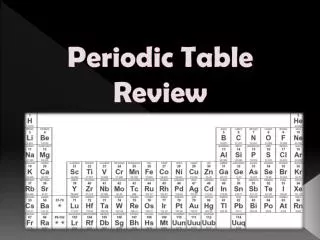
Information from Periodic Table • Atomic number • Chemical symbol • Atomic mass • Chemical name • Plus countless other information, depending on the table used!
Columns of Table = groups or families • Newer designation: 1-18 • 8 main families labeled with Roman Numeral and letter ‘A’ • Elements within the same family have similar but not identical properties • Electron configuration varies periodically (pattern of valence electrons) • family # 1-2-13-14-15-16-17-18 • or IA-IIA-IIIA-IVA-VA-VIA-VIIA-VIIIA • valence e- 1-2-3-4-5-6-7-8
16 18 1 2 15 17 14 13 GROUPS/FAMILIES GIVES # VALENCE E-
Definition • Valence electrons • electrons found in the outermost energy level • use the group number to determine number of valence electrons
Group 1 or IA Group 2 or IIA Group13 or IIIA Group 14 or IVA Group 15 or VA Group 16 or VIA Group 17 or VIIA Group 18 or VIIIA Groups 3-12 Lanthanide & Actinide series Alkali metals Alkaline Earth metals Boron family Carbon family Nitrogen family Chalogens Halogens Noble gases Transition metals Inner transition metals Names of Families
Rows of Table = periods • Properties are not alike within a period • 7 periods = number of energy levels in atom • 2 additional rows at bottom of table are Lanthanide and Actinide series and belong to periods 6 and 7
PERIOD = # ENERGY LEVELS 1 2 3 4 5 6 7
Trends • Valence number • Charge of ion • Tendency to gain or lose electrons • Atomic size (radius) • Ionization energy • Electron affinity • Electronegativity • Reactivity • Metallic vs. nonmetallic properties
The physical and chemical properties of the elements are periodic functions of their atomic number. The tendency of atoms to gain or lose electrons so they acquire 8 electrons in their outer energy level for stability Octet Rule PeriodicLaw
energy required to remove an electron from an atom; to form a cation kJ/mol attraction of an atom for an added electron; to form an anion kJ/mol Ionization Energy Electron Affinity
estimate of the size of an atom distance between the nucleus and the likely location of electron cloud ability of an atom to compete with other atoms for electrons shared between them i.e. who wants them more? Atomic Radius Electro-negativity
Across a Period • radius decreases • ionization energy increases • electronegativity increases • b/c • number of protons increases, as does number of electrons… • number of valence electrons increases… • so EMF increases… • so valence electrons held more tightly…
Atomic Radius PERIOD 3 PERIOD 2
Ionization Energy PERIOD 2 PERIOD 3
Electronegativity PERIOD 2 PERIOD 3
Down a Family • radius increases • ionization energy decreases • electronegativity decreases • b/c • number of energy levels increases… • so valence electrons held less tightly b/c further away from nucleus…
Atomic Radius Family IA Family IIA
Ionization Energy Family IIA Family IA
Electronegativity Family IIA Family IA
Alkaline Earth Metals Lab • An element that is highly reactive tends to gain or lose electrons very easily. • A precipitate is an insoluble solid formed as a result of a chemical reaction. • In this lab, magnesium formed the least number of precipitates, while barium formed the greatest number of precipitates. • Why do you think reactivity increases down a family???
REGIONS OF THE TABLE One more! NONMETALS METALLOIDS METALS
Metals • Found to LEFT of zigzag line on table • Good conductors of heat & electricity • Malleable (can hammer into thin sheet without breaking) • Ductile (can pull into thin wire without breaking) • High melting & boiling points • Shiny in luster • Tend to lose electrons
Nonmetals • Found to RIGHT of zigzag line on table • Poor conductors of heat & electricity • Brittle when solid (break irregularly) • Dull in luster • Low melting & boiling points • Tend to gain electrons
Metalloids aka Semi-Metals • Found along both sides of zigzag line, with exception of Al and Po • Properties of both metals & nonmetals • Okay conductors of heat & electricity • Shiny or dull
Who am I? • Use the clues and a periodic table to predict the element. • The number of clues is shown in the lower right corner.
1. Who am I? • Very good ability to conduct electricity • Never found alone in nature • When combined with other elements, usually give up 1 valence electron • Only one letter in symbol 4 clues
2. Who am I? • More valence electrons than oxygen • More protons than calcium • Fewer protons than krypton 3 clues
3. Who am I? • Poor conductor • Usually found as a gas • Do not bond well with others • Sometimes found in signs outside restaurants 4 clues
4. Who am I? • Second lowest electronegativity of period • One of most reactive in family • Not radioactive • Add 50 protons and I’m now another element in my family 4 clues
5. Who am I? • Usually found as a gas • Very reactive • Lightest element in family • Often form -1 ion 4 clues
6. Who am I? • Radioactive • Highest atomic radius in family • Usually form -2 ion 3 clues
7. Who am I? • Never found alone or unbonded in nature • Most commonly form +2 ion • Second highest number of protons in family 3 clues
8. Who am I? • All other members of family are metals • Most abundant element in universe 2 clues
9. Who am I? • Metals, nonmetals, and semi-metals all found in family • Same number of protons as the sum of the protons in the two elements directly above 2 clues
10. Who am I? • Highest ionization energy in family • Family contains metals, nonmetals, and semi-metals • Can form positive or negative ion 3 clues
That’s All Folks! Once you have finished, check your answers with a peer.