Classification of Matter
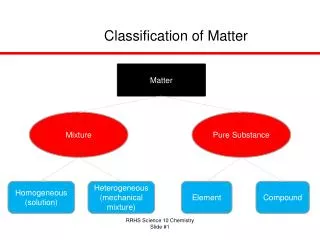
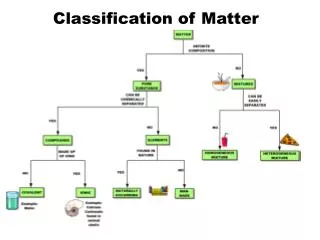
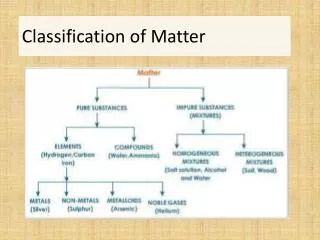
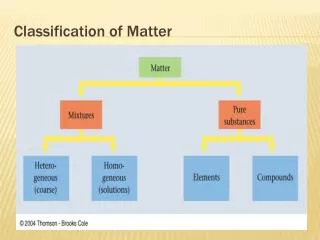
Classification of Matter. Matter. Mixture. Pure Substance. Homogeneous (solution). Heterogeneous (mechanical mixture). Element. Compound. Classification of Matter (Alternate). Matter. Homogeneous. Heterogeneous. Mixture. Pure Substance. Element. Compound. Physical properties.

Classification of Matter
E N D
Presentation Transcript
Classification of Matter Matter Mixture Pure Substance Homogeneous (solution) Heterogeneous (mechanical mixture) Element Compound
Classification of Matter (Alternate) Matter Homogeneous Heterogeneous Mixture Pure Substance Element Compound
Physical properties • A physical property is any aspect of matter that can be measured or seen without changing the composition of the matter. • Qualitative • Odor, color, texture, state, malleability • Quantitative • Melting point, boiling point, density, mass
Physical Change • Does not change the composition of the matter - doesn't change what the substance is • In a mixture, individual components retain properties of the original mixture; e.g. dissolving salt in water • Change of state (melting, freezing, etc.) is an example of a physical change
Physical Separation • Separating a mixture based on physical properties • Involves a physical change only (not chemical) • Individual components have properties of the mixture • Examples: • Filtration • Using magnetic properties • Sedimentation, using density differences
Chemical Properties • A chemical property is any property of matter that becomes evident during a chemical reaction. • Can only be measured by changing a substance's chemical identity. • Chemical properties cannot be determined just by viewing or touching the matter.
Chemical Change • A new substance is formed and energy is either given off or absorbed • Involves energy • If heat is given off during the reaction, than the reaction is considered to be exothermic. • If heat is required for the reaction, than the reaction is considered to be endothermic. • Composition of the substance is altered • New substances are produced with properties different from the original substance • Not easily reversed
Evidence of Chemical Change • The following may indicate that a chemical change has occurred • Colour Change • Temperature Change • Odour Given Off • Precipitate is Formed • Gas Produced • Any new substance produced • Examples • Burning • Metal in acid • Electrolysis of water
Chemical Separation • Separating a substance using a chemical change • Can be used to separate compounds