Theory
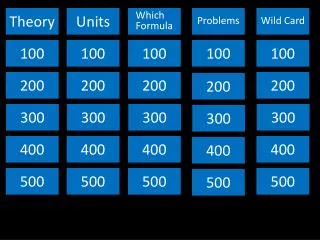
Theory. Units. Which Formula. Problems. Wild Card. 100. 100. 100. 100. 100. 2 00. 2 00. 2 00. 2 00. 2 00. 300. 300. 300. 300. 300. 400. 400. 400. 400. 400. 5 00. 5 00. 5 00. 5 00. 5 00. Theory 100. Gas pressure is caused by:

Theory
E N D
Presentation Transcript
Theory Units Which Formula Problems Wild Card 100 100 100 100 100 200 200 200 200 200 300 300 300 300 300 400 400 400 400 400 500 500 500 500 500
Theory 100 • Gas pressure is caused by: A - gas molecules reacting with other gas molecules B - gas molecules hitting the walls of a container C - gas molecules heating up D - gas molecules hitting other gas molecules ANSWER
Theory 100 • Gas pressure is caused by: B - gas molecules hitting the walls of a container Main
Theory 200 • Each of these flasks contains the same number of gas molecules. In which would the pressure be highest? • A - Flask 1 • B - Flask 4 • C - Flask 3 • D - Flask 2 ANSWER
Theory 200 • Each of these flasks contains the same number of gas molecules. In which would the pressure be highest? • B - Flask 4 Main
Theory 300 • At a temperature of 25°C and a pressure of 1atm, which of the following molecules will effuse the most rapidly? • A - oxygen, O2 • B - carbon dioxide, CO2 • C - hydrogen chloride, HCl • D - nitrogen, N2 ANSWER
Theory 300 • At a temperature of 25°C and a pressure of 1atm, which of the following molecules will effuse the most rapidly? • D - nitrogen, N2 Main
Theory 400 • As the temperature of the gas in a balloon decreases, which of the following occurs? • A – The average kinetic energy of the gas decreases • B – The gas pressure inside the balloon increases • C – The volume of the balloon increases • D – All of the above ANSWER
Theory 400 • As the temperature of the gas in a balloon decreases, which of the following occurs? • A – The average kinetic energy of the gas decreases Main
Theory 500 • At a certain temperature and pressure, 2 mol of carbon dioxide has a volume of 3L. A 3L sample of hydrogen at the same temperature and pressure___. • A – contains the same number of atoms • B – has a higher density • C – contains the same number of molecules • D – has the same mass ANSWER
Theory 500 • At a certain temperature and pressure, 2 mol of carbon dioxide has a volume of 3L. A 3L sample of hydrogen at the same temperature and pressure___. • C – contains the same number of molecules Main
Units 100 Convert to Kelvin: 15°C ANSWER
Units 100 Convert to Kelvin: 15°C 15 + 273 = 288K Main
Units 200 Convert to Celsius: 100K ANSWER
Units 200 Convert to Celsius: 100K 100-273 = -173°C Main
Units 300 In the ideal gas law, what is the value of R? ANSWER
Units 300 In the ideal gas law, what is the value of R? R = 0.0821 L•atm/K•mol Main
Units 400 Convert to atm’s: 123.4 kPa Answer
Units 400 Convert to atm’s: 123.4 kPa 123.4 kpa x (1atm/101.325kpa) = 1.218 atm Main
Units 500 What is standard pressure and temperature? Answer
Units 500 What is standard pressure and temperature? T = 273K P = 1atm Main
Which Formula 100 • A sample of nitrogen occupies a volume of 250 ml at 25°C. What volume will it occupy at 95°C? Answer
Which Formula 100 • A sample of nitrogen occupies a volume of 250 ml at 25°C. What volume will it occupy at 95°C? • Charles’ Law: V1/T1 = V2/T2 Main
Which Formula 200 • A 175 ml sample of neon gas had its pressure changed from 75 kPa to 150 kPa. What is the new volume? Answer
Which Formula 200 • A 175 ml sample of neon gas had its pressure changed from 75 kPa to 150 kPa. What is the new volume? Boyle’s Law: P1 x V1 = P2 x V2 Main
Which Formula 300 • A rigid container of O2 has a pressure of 185 kPa at a temperature of 458 K. What is the pressure at 283 K? Answer
Which Formula 300 • A rigid container of O2 has a pressure of 185 kPa at a temperature of 458 K. What is the pressure at 283 K? • Gay Lussac’sLaw : P1 / T1 = P2 / T2 Main
Which Formula 400 • Chlorine gas exerts a pressure of 900 torr. When the pressure is changed to 1.35 atm, it’s volume is 250 ml. What was the original volume? Answer
Which Formula 400 • Chlorine gas exerts a pressure of 900 torr. When the pressure is changed to 1.35 atm, it’s volume is 250 ml. What was the original volume? Gay-Lassac’s: P1 x V1 = P2 x V2 Main
Which Formula 500 • What is the volume occupied by 95 grams of chlorine gas at STP? Answer
Which Formula 500 • What is the volume occupied by 95 grams of chlorine gas at STP? Ideal: PV = nRT Main
Problems 100 • How many moles of nitrogen are in a flask with a volume of 150.0 ml at a pressure of 200.0 kPa and a temperature of 250 K Answer
Problems 100 • How many moles of nitrogen are in a flask with a volume of 150.0 ml at a pressure of 200.0 kPa and a temperature of 250 K n = .014 moles Main
Problems 200 A small sample of helium gas occupies 6 mL at a temperature of 250 K. At what temperature does the volume expand to 9 mL? Answer
Problems 200 A small sample of helium gas occupies 6 mL at a temperature of 250 K. At what temperature does the volume expand to 9 mL? 375 K Main
Problems 300 When a supply of hydrogen gas is held in a 4.00 liter container at 320 K it exerts a pressure of 1.05 atm. The supply is moved to a 2.00 liter container, and cooled to 160 K. What is the new pressure of the confined gas. Answer
Problems 300 When a supply of hydrogen gas is held in a 4.00 liter container at 320 K it exerts a pressure of 1.05 atm. The supply is moved to a 2.00 liter container, and cooled to 160 K. What is the new pressure of the confined gas P = 1.05 atm Main
Problems 400 A sample of helium gas diffuses at .0659 m/s at a certain temperature. Calculate the velocity of chlorine gas at the same temperature. Answer
Problems 400 A sample of helium gas diffuses at .0659 m/s at a certain temperature. Calculate the velocity of chlorine gas at the same temperature. V2 = .0221 m/s Main
Problems 500 A mixture of gases at a total pressure of 95 kPa contains N2, CO2 and O2. The partial pressure of CO2 is 24 kPa, and the partial pressure of N2 is 35 kPa. What is the pressure of O2 in atms? Answer
Problems 500 A mixture of gases at a total pressure of 95 kPa contains N2, CO2 and O2. The partial pressure of CO2 is 24 kPa, and the partial pressure of N2 is 35 kPa. What is the pressure of O2 in atms? .36 atm Main
Wild Card 100 As the temperature of a fixed volume of gas increases, the pressure will ______. Answer
Wild Card 100 As the temperature of a fixed volume of gas increases, the pressure will ______. • Increase Main
Wild Card 200 .A gas occupies a volume of 120. ml at 25.0°C and 97.0 kPa. What is the volume of the gas at STP? Answer
Wild Card 200 .A gas occupies a volume of 120. ml at 25.0°C and 97 kPa. What is the volume of the gas at STP? V2 = .105L Main
Wild Card 300 Given the following chemical equation: CH4+ 2O2 -> CO2 + 2H2O What volume of steam H2O will be produced at 35°C and 2.6 atm if .875 mol of CH4 completely react with excess oxygen? Answer
Wild Card 300 Given the following chemical equation: CH4+ 2O2 -> CO2 + 2H2O What volume of steam H2O will be produced at 35°C and 2.6 atm if .875 mol of CH4 completely react with excess oxygen? 17L Main
Wild Card 400 The volume of a gas is tripled while the temperature is held constant. How does the gas pressure change? A – It is also tripled B – It is reduced by one third C – It does not change D – It depends on the type of gas Answer
Wild Card 400 The volume of a gas is tripled while the temperature is held constant. How does the gas pressure change? B – It is reduced by one third Main
Wild Card 500 A 15.50 gram sample of a gas exerts a pressure of 1.40 atmospheres when held in an 8.00 liter at 22 °C. What is the molar mass of the gas? Answer