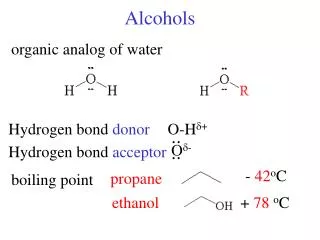
Alcohols
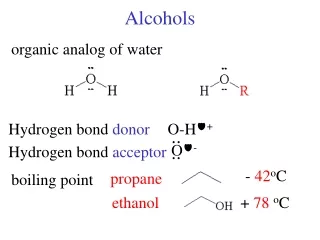
Alcohols. Hydrogen Bonding. Three ethanol molecules. Hydrogen Bonding & boiling point. Increases boiling point, higher temperature needed to separate the molecules. Hexane 69 deg. 1-pentanol 138 1,4-butanediol 230 .

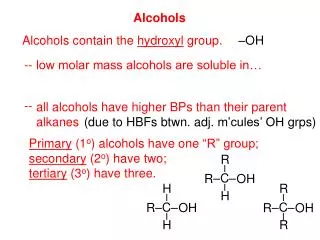
Alcohols
E N D
Presentation Transcript
Hydrogen Bonding Three ethanol molecules.
Hydrogen Bonding & boiling point Increases boiling point, higher temperature needed to separate the molecules. Hexane 69 deg. 1-pentanol 138 1,4-butanediol 230 Ethanol 78 deg Dimethyl ether 24
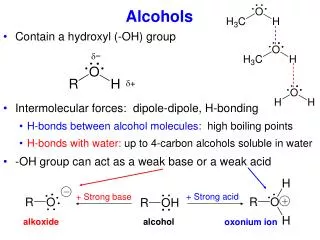
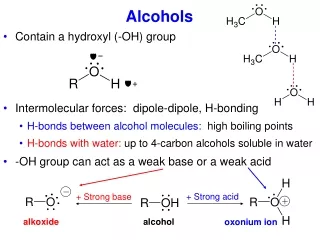
Earlier Discussion of Acidity Increasing Hinderance of Solvation RO-H RO – (solvated) + H + (solvated) Alkoxide ion, base Methanol Ethanol 2-Propanol 2-Methyl-2-propanol Increasing Basicity of Alkoxide Anion, the conjugate base Increasing Acidity of the alcohol Alkoxides can be produced in several ways… Recall: H2O + Na Na+ + OH- + ½ H2(g) Alkoxide, strong base, strong nucleophile (unless sterically hindered) Alcohols behave similarly ROH + Na Na+ + OR- + ½ H2(g) Also: ROH + NaH Na+ + OR- + ½ H2(g)
-OH as a Leaving Group Poor leaving group, hydroxide ion. R-OH + H + R-OH2+ Protonation of the alcohol sets-up a good leaving group, water. Another way to turn the –OH into a leaving group…
Conversion to Alkyl Halide,HX + ROH RX + H2O When a carbocation can be formed (Tertiary, Secondary alcohols) beware of rearangement. SN1 Expect both configurations. When a carbocation cannot be formed. Methanol, primary. SN2
But sometimes experiment does not agree with our ideas… Observed reaction • The problem: • Rearrangement of carbon skeleton which usually indicates carbocations. • Reacting alcohol is primary; do not expect carbocation. • Time to adjust our thinking a bit…. Not a primary carbocation
Other ways to convert: ROH RX We have used acid to convert OH into a good leaving group There are other ways to accomplish the conversion to the halide. Leaving group. Leaving group. Next, a very useful alternative to halide…
An alternative to making the halide: ROH ROTs Preparation from alcohols. p-toluenesulfonyl chloride Tosyl chloride TsCl Tosylate group, -OTs, good leaving group, including the oxygen. The configuration of the R group is unchanged.
Example Preparation of tosylate. Retention of configuration
Substitution on a tosylate The –OTs group is an excellent leaving group
Acid Catalyzed Dehydration of an Alcohol, discussed earlier as reverse of hydration Secondary and tertiary alcohols, carbocations Protonation, establishing of good leaving group. Elimination of water to yield carbocation in rate determining step. Expect tertiary faster than secondary. Rearrangements can occur. Elimination of H+ from carbocation to yield alkene. Zaitsev Rule followed.
Primary alcohols • Problem: primary carbocations are not observed. Need a modified, non-carbocation mechanism. • Recall these concepts: • Nucleophilic substitution on tertiary halides invokes the carbocation but nucleophilic substitution on primary RX avoids the carbocation by requiring the nucleophile to become involved immediately. • The E2 reaction requires the strong base to become involved immediately. Note that secondary and tertiary protonated alcohols eliminate the water to yield a carbocation because the carbocation is relatively stable. The carbocation then undergoes a second step: removal of the H+. The primary carbocation is too unstable for our liking so we combine the departure of the water with the removal of the H+. What would the mechanism be???
Here is the mechanism for acid catalyzed dehydration of Primary alcohols 1. protonation 2. The carbocation is avoided by removing the H at the same time as H2O departs (like E2). As before, rearrangements can be done while avoiding the primary carbocation.
Principle of Microscopic Reversibility Same mechanism in either direction.
Pinacol Rearrangement: an example of stabilization of a carbocation by an adjacent lone pair. Overall:
Mechanism Reversible protonation. Elimination of water to yield tertiary carbocation. This is a protonated ketone! 1,2 rearrangement to yield resonance stabilized cation. Deprotonation.
Oxidation Primary alcohol Na2Cr2O7 Na2Cr2O7 RCH2OH RCH=O RCO2H Na2Cr2O7 (orange) Cr3+ (green) Actual reagent is H2CrO4, chromic acid. Secondary Na2Cr2O7 KMnO4 (basic) can also be used. MnO2 is produced. R2CHOH R2C=O Tertiary The failure of an attempted oxidation (no color change) is evidence for a tertiary alcohol. R3COH NR
Oxidation using PCC Primary alcohol Stops here, is not oxidized to carboxylic acid PCC RCH2OH RCH=O Secondary PCC R2CHOH R2C=O
Mechanistic Notes Cyclic structure is formed during the reaction. Evidence of cyclic intermediate.
Sulfur Analogs, Thiols Preparation RI + HS- RSH SN2 reaction. Best for primary, ok secondary, not tertiary (E2 instead) Oxidation • Acidity • H2S pKa = 7.0 • RSH pKa = 8.5