Ethers and Epoxides
Ethers and Epoxides. By Dr. Nahed Nasser. THE CHEMISTRY OF Ethers and Epoxides. CONTENTS Structure and classification of ethers Nomenclature of ethers Physical properties of ethers Preparation of ethers from alcohols and alkyl halides

Ethers and Epoxides
E N D
Presentation Transcript
Ethers and Epoxides By Dr. Nahed Nasser
THE CHEMISTRY OF Ethers and Epoxides • CONTENTS • Structure and classification of ethers • Nomenclature of ethers • Physical properties of ethers • Preparation of ethers from alcohols and alkyl halides • Reactions of opened ethers with HI • Synthesis of epoxides (3-memebered cyclic ethers) from alkenes • Reactions of epoxides with H2O, ROH, HX, LiAlH4, phenol, Grignard reagent and NH3
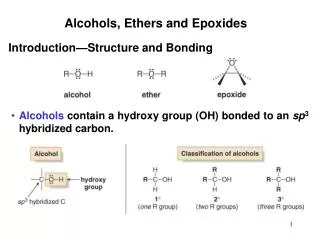
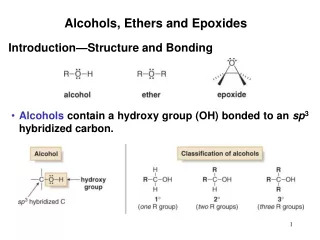
STRUCTURE OF ETHERS • Ether:is a class of organic compounds that contain an ether group — an oxygenatom connected to two alkyl or aryl groups via σ bonds. thus Ethers feature C-O-C linkage — of general formula R–O–R • Types of ethers • Symmetrical ethers : in that the two substituents are the same i.e. R-O-R or Ar-O-Ar • Unsymmetrical ethers are those have two different substituents i.e. R-O-R’ or R-O-Ar or Ar-O-Ar’ • Nomeclature OF Ethers • Common names • The common names of ethers are derived by naming the alkyl groups bonded to the oxygen then listing them in alphabetical order followed by the word "ether". • IUPAC names • The ether functional group does not have a characteristic IUPAC nomenclature suffix, so it is necessary to designate the smaller alkyl group as an alkoxy substituent of a parent compound ( alkane , or alkene, or alkyne,or alcohol, .)
The common alkoxysubstituents are given names derived from their alkyl component,
CommonDimethyl ether Diethyl ether Divinyl ether Diphenyl ether IUPACMethoxy methane Ethoxy ethane Phenoxy benzene CommonHexyl methyl ether Ethyl methyl ether Methyl vinyl ether Methyl Phenyl ether (anisole) IUPAC Methoxy hexane methoxyethane MethoxyetheneMethoxy benzene IUPAC 3-Methoxyhexane 5-Ethoxy-2-heptene Propenyloxy benzene
Physical Properties Solubility : Ethers are polar compounds because of the polar nature of both the C-O bonds due to the high electronegativity of the O atom compared to carbon atom. Thus they can be involved in H-bonding with systems able to donate H (e.g. water). Consequently their solubility in aqueous media is similar to analogous alcohols. Boiling point : An ether cannot form hydrogen bonds with other ether molecules since there is no H to be donated resulting in lower melting and boiling points compared to analogous alcohols. A hydrogen bond is the attractive interaction of a hydrogen atom with an electronegative atom, like nitrogen, oxygen or fluorine. The hydrogen must be covalently bonded to another electronegative atom to create the bond. These bonds can occur between molecules (intermolecularly), or within different parts of a single molecule (intramolecularly).. Intermolecular hydrogen bonding is responsible for the high boiling point of water (100 °C). Q-Ethanol C2H5OH and methyl ether CH3OCH3 have the same weight. Which has a higher boiling point?
Preparation of Ethers 1- Dehydration of alcohols: 2- The Williamson synthesis :
Cyclic Ethers IUPAC OxiraneOxetaneOxlaneOxane1,4-Dioxane CommonEthylene oxide Trimethyleneoxide TetrahydrofuraneTetrahydropyrane Preparation of cyclic ethers