Chapter 7: Cellular Respiration
Chapter 7: Cellular Respiration. Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display. Overview of Cellular Respiration. Cellular respiration -the step-wise release of energy from carbohydrates and synthesis of ATP requires oxygen (O 2 )

Chapter 7: Cellular Respiration
E N D
Presentation Transcript
Chapter 7: Cellular Respiration Copyright © The McGraw-Hill Companies, Inc. Permission required for reproduction or display.
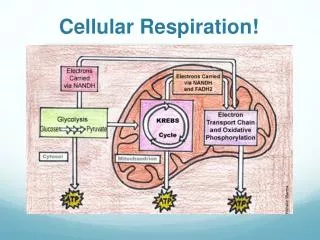
Overview of Cellular Respiration • Cellular respiration -the step-wise release of energy from carbohydrates and synthesis of ATP • requires oxygen (O2) • releases carbon dioxide (CO2) • glucose split to carbon dioxide and water.
oxidation of glucose (exergonic)drives ATP synthesis(endergonic), coupled reaction one glucose molecule yields 36 to 38 ATP molecules (efficiency of approx. 40%)
NAD+ and FAD • Nicotinamide adenine dinucleotide), flavin adenine dinucleotide • Coenzymes • NAD+ used more often • accept two electrons plus a hydrogen ion (H+) Reduced or oxidized?
Phases of Complete Glucose Breakdown • The oxidation of glucose by removal of hydrogen atoms involves four phases: • Glycolysis – the breakdown of glucose to two molecules of pyruvate in the cytoplasm; no oxygen needed; yields 2 ATP • Transition reaction – pyruvate is oxidized to a 2-carbon acetyl group carried by CoA (acetyl CoA); CO2 is removed; twice per glucose molecule
Citric acid cycle – a cyclical series of oxidation reactions that give off CO2 produce one ATP, NADH, FADH; occurs twice per glucose molecule Electron transport system – a series of carriers that accept electrons removed from glucose and pass them from one carrier to the next until the final receptor, O2 is reached; water is produced; energy is released and used to synthesize 32 to 34 ATP If oxygen is not available, fermentation occurs in the cytoplasm instead of proceeding to cellular respiration.
Outside the Mitochondria: Glycolysis • Glycolysis occurs in the cytoplasm and is the breakdown of glucose to two pyruvate molecules. • Glycolysis is found in all organisms • Glycolysis does not require oxygen.
Energy-Investment Steps • two ATP are used to activate glucose, • Glucose splits into two C3 molecules (PGAL). • PGAL carries a phosphate group from ATP. • From this point on, each C3 molecule undergoes the same series of reactions.
Energy-Harvesting Steps • Oxidation of PGAL now occurs by the removal of electrons that are accompanied by hydrogen ions, both picked up by the coenzyme NAD+: • 2 NAD+ + 4H → 2 NADH + 2 H+ • The oxidation of PGAL and subsequent substrates results in four high-energy phosphate groups used to synthesize ATP in substrate-level phosphorylation.
Inputs: Glucose 2 NAD+ 2 ATP 4 ADP + 2 P Outputs: 2 pyruvate 2 NADH 2 ADP 2 ATP (net gain) Glycolysis summary
Oxygen present - pyruvate enters the mitochondria. If oxygen is not available, fermentation occurs Fermentation - anaeorbic (does not require oxygen), in humans lactic acid is produced.
Review • mitochondrion – • double membrane • intermembrane space between the two layers. • Cristae -folds of inner membrane that jut out into the matrix, the innermost compartment • transition reaction and citric acid cycle occur in the matrix • the electron transport system is located in the cristae.
Transition Reaction • 1) connects glycolysis to the citric acid cycle • 2) Pyruvate is converted to a C2acetyl group attached to coenzyme A (together called acetyl CoA) • 3) CO2 is released. • 4) NAD+ is converted to NADH + H+
Citric Acid Cycle • 1) cyclical metabolic pathway located in the matrix of the mitochondria. • 2) acetylCoA joins a C4 molecule, and C6citrate results. • AcetylCoA will be oxidized to CoA and 2 CO2 molecules.
4) oxidation occurs when NAD+ accepts electrons (happens 3 times) and FAD accepts electrons once. Gain 1 ATP per acetyl CoA (substrate-level phosphorylation)
Inputs: 2 acetyl groups 6 NAD+ 2 FAD 2 ADP + 2 P Outputs: 4 CO2 6 NADH 2 FADH2 2 ATP Citric acid cycle inputs and outputs per glucose molecule
Electron Transport System • located in the cristae of mitochondria • series of protein carriers (some are cytochromes), pass electrons from one to the other. • 3) electrons removed from NADH and FADH2 and enter the electron transport system. • pair of electrons is passed from carrier to carrier • energy is released and used to form ATP molecules by (oxidative phosphorylation)
6) Oxygen receives energy-spent electrons at the end of the electron transport system. 7) oxygen combines with hydrogen, and water forms: Where did the hydrogen come from?
Organization of Cristae • electron transport system is located in the cristae • consists of protein complexes and mobile carriers. • The carriers use the energy released by electrons as they move down the carriers to pump H+ from the matrix into the intermembrane space of the mitochondrion.
pH gradient is established with few H+ in the matrix and many in the intermembrane space. Is the pH of the matrix high or low? Is the pH of the intermembrane space high or low? cristae contain an ATP synthase complex through which hydrogen ions Which way do they flow? Energy used to synthesis ATP (chemiosmosis)
Advantages and Disadvantages of Fermentation • Fermentation can provide a rapid burst of ATP in muscle cells, even when oxygen is in limited supply. • Lactate, however, is toxic to cells. • Initially, blood carries away lactate as it forms; eventually lactate builds up, lowering cell pH, and causing muscles to fatigue. • Oxygen debt occurs, and the liver must reconvert lactate to pyruvate.
Efficiency of Fermentation • Two ATP produced during fermentation are equivalent to 14.6 kcal; complete oxidation of glucose to CO2 and H2O represents a yield of 686 kcal per molecule of glucose. • Thus, fermentation is only 2.1% efficient compared to cellular respiration. • (14.6/686) x 100 = 2.1%
Metabolic Pool and Biosynthesis • Degradative reactions, which occur in catabolism, break down molecules and are exergonic. • Synthetic reactions, which occur during anabolism, tend to be endergonic. • Catabolism drives anabolism because catabolism results in ATP buildup used by anabolism.
Catabolism • Molecules aside from glucose can enter the catabolic reactions of cellular respiration. • When a fat is used for energy, it breaks down into glycerol and three fatty acids; glycerol is converted to PGAL, and the fatty acids are converted to acetyl-CoA, thus both types of molecules can enter the citric acid cycle.
The carbon backbones of amino acids can also enter the reactions of cellular respiration to provide energy. The amino acid first undergoes deamination, or the removal of the amino group; the amino group becomes ammonia (NH3) and is excreted. Where the carbon portion of the amino acid enters the reactions of respiration depends on its number of carbons.
Anabolism • The substrates of the pathways of cellular respiration can also be used as starting materials for synthetic reactions. • This is the cell’s metabolic pool, in which one type of molecule can be converted into another. • In this way, dietary carbohydrates can be converted to stored fat, and come substrates of the citric acid cycle can be transaminated into amino acids.
Chapter Summary • During cellular respiration, glucose is oxidized to CO2 and H2O; this exergonic reaction drives ATP buildup. • Four phases of cellular respiration occur: • 1) Glycolysis, in the cytosol, is the breakdown of glucose to two pyruvates, with the formation of 2 NADH and net gain of 2 ATP. • 2) A transition reaction takes place to convert pyruvate into acetyl-CoA, with CO2 given off; two NADH result in total.
3) The acetyl group enters the citric acid cycle, located in the matrix of the mitochondria; complete oxidation follows, and two CO2, three NADH, one FADH2, and two ATP are formed – the entire cycle runs twice per glucose molecule. 4) The final stage of glucose breakdown is the electron transport system located in the cristae of the mitochondria; electrons from NADH and FADH2 are passed down a chain of carriers until O2 is reached and H2O is formed. ATP is formed during oxidative phosphorylation via chemiosmosis.
Fermentation involves glycolysis, followed by the reduction of pyruvate to lactate or alcohol and CO2; in humans, it provides a quick burst of energy but triggers oxygen debt. Carbohydrate, protein, and fat can be used for energy, and their components can be used for synthesis of needed compounds; both anabolism and catabolism use the same metabolic pool of reactants.