Nitrogen and Ecosystem Nutrient Cycling
190 likes | 468 Views
Nitrogen and Ecosystem Nutrient Cycling. Nicole and Sarah Biogeochemistry of Northern Ecosystems March 2005. Cycling Essential Life Elements. Interactions between plants, animals, and soil microbes link the internal biogeochemistry of terrestrial ecosystems. Essential life elements:

Nitrogen and Ecosystem Nutrient Cycling
E N D
Presentation Transcript
Nitrogen and Ecosystem Nutrient Cycling Nicole and Sarah Biogeochemistry of Northern Ecosystems March 2005
Cycling Essential Life Elements • Interactions between plants, animals, and soil microbes link the internal biogeochemistry of terrestrial ecosystems. • Essential life elements: C, N, P, S, P, Ca, Mg, F -often limiting and their supply may control NPP • Biosphere exerts strong control on major life elements- intrasystem cycle is the annual cycle of nutrients in vegetation and is responsible for long-term retention of elements
Annual Nutrient Requirements for Growth (Schlesinger, 1997 p.168)
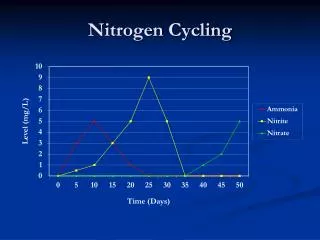
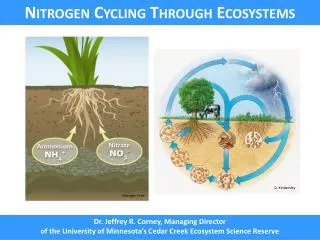
General Nitrogen Information • Out of those elements required to sustain life, Nitrogen has the greatest total abundance in the atmosphere, biosphere, and hydrosphere but it is the least readily available to sustain life. • In the form of molecular nitrogen, two nitrogen atoms are held together by a triple bond. Only a small number of N-fixing species have the energy required to break apart this bond. • There are two categories of Nitrogen in nature, N which is the non-reactive molecular form of nitrogen and Nr which is all the reactive nitrogen in the earths atmosphere and biosphere. • Reactive nitrogen was originally formed by one of two ways: lightning or biological fixation.
The Nitrogen Cycle Note: Nitrous oxide is N20.
Denitrification • Denitrification is the loss of Nr from the biosphere because of its conversion back to its molecular form, with a triple bond. • Three conditions required: 1) presence of NO3- or nitrate, 2) availability of labile organic matter, and 3) absence or low concentration oxygen. • The residence time of water is an important factor in controlling how much denitrification goes on. • Denitrification is an effective sink for N • Denitrification can control phytoplankton blooms and degree of eutrophication
Denitrification Reduces the Downstream N Transport (Galloway et al, 2003) Land 30-70% entering rivers denitrified Rivers 10-80% entering estuaries denitrified Estuaries Cont. Shelves >80% entering shelves denitrified ?
How Humans Have Impacted the Nitrogen Cycle • Three main causes of the global increase in Nr: • Cultivation of plants associated with n-fixers (e.g. legumes) increasing BNF • Industrial N Fixation by the Haber-Bosch process- (fertilizer and bombs) 1. Generate Hydrogen CH4+H2ONi catalystCO+3H2 React H2 + N2 to produce NH3 N2 + 3H22NH3 3. Fossil Fuel burning- converts non reactive N into NOx
billion x1012 g yr-1 Haber-Bosch World population Crop Fixation Fossil Fuel
Discussion Questions Hypothetical Situation • A farmer lives in close proximity to the watershed of a major river which feeds into the ocean. He uses nitrogen fertilizer to increase his crop yield. In an effort to become more eco-friendly he wants to know what effect his farm will have on the surrounding ecosystem and its nitrogen cycle? Suggest possible ways he can improve his farming practices and maintain economic stability. • Since the industrial revolution there has been a dependency on the burning of fossil fuels for energy. One of the effects of this process is the release of NOx to the atmosphere. How is NOx created and what are the implications of its presense in the atmosphere?
Discussion Questions- Potential Ecosystem Effects Agriculture Fossil Fuel • Initially release of NOx in troposphere • produces ozone (health implications) • Increases accumulation of small • particles in air- decreases atmospheric • visibility • Acid precipitation and N deposition • N2O emitted back to atmosphere can • first contribute to greenhouse warming • N2O in the stratosphere can deplete • ozone layer • Over saturation of N leads to high • N loss from system to soil, • atmosphere and water • Inputs of Nr exceeds the systems • ability to remove N through • denitrification • transported N can alter species • composition and trophic structure • in surrounding ecosystems • High levels of N can degrade • habitats (e.g. eutrophication, • areas of anoxia and hypoxia)
Nitrogen Saturation and Ecosystem Degradation • Inputs of N from the atmosphere in the northeastern U.S. are 5 to 20 times higher than those under pristine conditions (10-50kg N ha-1yr-1) • Added N may be accumulated in organic matter however, forest decline is • observed as the ecosystem becomes saturated with N
Discussion Questions- Potential Solutions Agriculture Fossil Fuel ?ideas ?ideas