Pascal’s Principle:
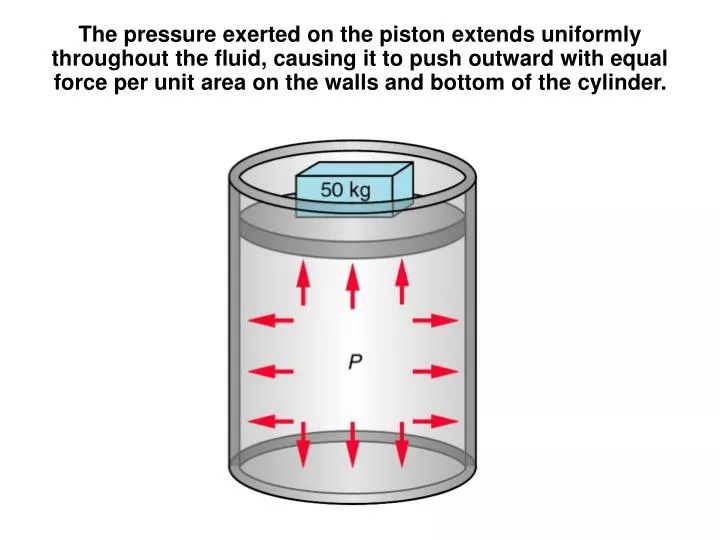
The pressure exerted on the piston extends uniformly throughout the fluid, causing it to push outward with equal force per unit area on the walls and bottom of the cylinder. Pascal’s Principle: Any change in the pressure of a fluid is transmitted uniformly in all directions throughout the fluid.

Pascal’s Principle:
E N D
Presentation Transcript
The pressure exerted on the piston extends uniformly throughout the fluid, causing it to push outward with equal force per unit area on the walls and bottom of the cylinder.
Pascal’s Principle: Any change in the pressure of a fluid is transmitted uniformly in all directions throughout the fluid.
A small force F1 applied to a piston with a small area produces a much larger force F2 on the larger piston. This allows a hydraulic jack to lift heavy objects.
Torricelli filled a tube with mercury and inverted it into an open container of mercury. Air pressure acting on the mercury in the dish can support a column of mercury 76 cm in height. How much does the atmosphere weigh? The same as 76 cm of mercury.
Archimedes’ Principle: The buoyant force acting on an object fully or partially submerged in a fluid is equal to the weight of the fluid displaced by the object.
The weight of a column of water is proportional to the volume of the column. The volume V is equal to the area A times the height h. Equilibrium…
Density = mass / volume 1 cm3 of water weighs 1 gram rwater = 1.0 gm/cm3 1 Liter of water = 1000 cm3 weighs 1000 grams = 1.0 kg
Lecture Quiz 20 - Question 1: A large metal tank is filled with high pressure of Helium, used to fill Helium balloons. Consider how much the tank weighs when it is full, and when empty. The weight is the same full or empty. The empty tank weighs less than the full tank. The tank weighs less when it is full, because Helium is lighter than air.
Lecture Quiz 20 - Question 2: One cubic centimeter of Aluminum weighs 2.7 gms (rAl=2.7 gms/cm3). The Al cube is placed in a beaker of water. The upward buoyant force on the cube equals the downward force (weight). The weight of the displaced water equals the weight of the cube. The weight of the cube is larger than the buoyant force. The cube floats, because Aluminum is lighter than water.
Lecture Quiz 20 - Question 3: A block of wood is added to a beaker of water. It floats. The beaker rests on a scale that reads its weight. There is no change in weight, because the buoyant force equals the weight of the water. The weight increases by the weight of the block. The weight increases by an amount less than the weight of the block.
The Fahrenheit and Celsius scales use different numerical values for the freezing and boiling points of water. The Celsius degree is larger than the Fahrenheit degree.
Temperature: For objects #1 and #2 which are brought in contact, heat will flow from object #2 into object #1 if T2 > T1. Fahrenheit: TF = (9/5)TC + 32 Celsius: TC = (5/9)(TF - 32) Kelvin: TK = TC + 273.2 Zero K = “absolute zero”; No physical system can get colder than this.
The pressure plotted as a function of temperature for different amounts of different gases. When extended backward, the lines all intersect the temperature axis at the same point.
One-hundred grams of room-temperature water (20°C) is more effective than 100 grams of room-temperature steel shot in cooling a cup of hot water.
Specific Heat Capacity: How much heat is needed to increase the temperature of one gram of water by one degree C? Exactly 1 Calorie. 1 cal = 4.186 J (energy) Hence, the specific heat capacity is: Water 1.0 cal/gm/C Ice 0.49 Steel 0.11 It takes much less heat to raise the temperature of 1 gram of steel than 1 gram of water. Steel will not remove as much heat from your hot coffee; water is a better cooling agent.
What is heat? It is the energy that flows from a hot object to a cooler one.
Heat flows from the hotter object to the colder one when two objects at different temperature are placed in contact. The changes in temperature that result depend on the quantities of material and the specific heat capacity of each object.
Adding heat to a mixture of water and ice at 0°C melts the ice without changing the temperature of the mixture.
Will adding heat to a body always raise its temperature? No, there can be a change of phase. It takes 0.49 cal to raise 1 gram of ice from -1 C to 0 C. But, it takes another 80 cal to go from ice at 0 C to water at 0 C. Similarly, it takes 540 cal to go from 1 gram of water at 100 C to one gram of steam at 100 C.
Figure 10.12A falling mass turns a paddle in an insulated beaker of water in this schematic representation of Joule’s apparatus for measuring the temperature increase produced by doing mechanical work on a system.
An object can get hotter by • adding heat to it • doing work on it! • Total energy is conserved. Heat is another form of energy.