Stellar Nucleosynthesis
Stellar Nucleosynthesis. Charles Hyde 2 March 2009. Nucleosynthesis in Stars. Great triumphs of 20th century physics Discovery that sun, stars are mostly H Explanation of nuclear fusion reactions powering sun Nuclear Binding Energy Quantum mechanics Weak interaction ( beta decay)

Stellar Nucleosynthesis
E N D
Presentation Transcript
Stellar Nucleosynthesis Charles Hyde 2 March 2009
Nucleosynthesis in Stars • Great triumphs of 20th century physics • Discovery that sun, stars are mostly H • Explanation of nuclear fusion reactions powering sun • Nuclear Binding Energy • Quantum mechanics • Weak interaction ( beta decay) • Neutrino flux from sun, • Ray Davis, BNL (Cl detector in Homestake mine, SD) • Neutrinos from SuperNova • Kamiokande Water Cerenkov detector: SN1987a
Nucleosynthesis in the 21st Century • How exactly do Supernovae explode • Complex 3D hydrodynamics • Role of neutrino mass and oscillation? • See next 10 years of accelerator and reactor based neutrino experiments • Exact pathways for nucleosynthesis of elements heavier than Fe • FRIB project launched at MSU, Dec 2008 • Exact understanding of nuclear masses from fundamental theory • Quark mass - Higgs mechanism? FermiLab, LHC • Proton mass, nuclear force from QCD • Jlab, Lattice QCD, …
Nuclear Energy in Stars • E=mc2 • Release of energy by fusion reactions • Pp chain • CNO chain • Explosive nucleosynthesis
Stellar Hydrodynamics • See Lectures, Prof. Bueltmann • Astrophysics in a Nutshell, Section 3.1 • Temperature, Luminosity of sun from quantum black body emission • Hydrostatic/Gravity equilibrium determines temperature / pressure profile of sun. • Only mildly effected by profile of nucleo-synthesis (concentrated at temperature/pressure maximum at center)
Solar Properties • Temperature • Surface T=5800K, kT = 0.5eV • Center 15106 K. • Mass 21030 kg. • Radius R=7108 m. • Density • Average = 1.4 g/cm3. • Central = 150 g/cm3. • Total Luminosity 71033 W. • Gravitational potential energy released in collapse from infinity to R • GM2/(2R) = 21041 J.
Possible Source of Solar Luminosity • Gravitational collapse • Lifetime = [Potential Energy]/Luminosity • T = [21041 J] / [71033 W] = 1 yr • Nuclear Fusion • Enough energy • Slow enough to sustain sun for > 109 years • Reaction rate depends upon subtle details of nuclear physics
Nuclear Energy • E=mc2. • Atomic mass unit (u) • 1 mole of 12C defined to have a mass of 12 g • (This defines Avogadro’s number = 6.021023) • 1 neutral 12C atom has a mass = 12u • 1 u = (0.012 kg)c2/(12NA)=1.510-10 J • 1eV = (1.610-19 C)(1V)= 1.610-19 J • 1 u = 934106 eV • Nuclear Wallet cards www.nndc.bnl.gov/wallet/ • M(H) = 1u+ 8.071 MeV • M(n) = 1u+ 7.289 MeV • Mass(AZ) = A(1u)+(AZ) • (12C)=0
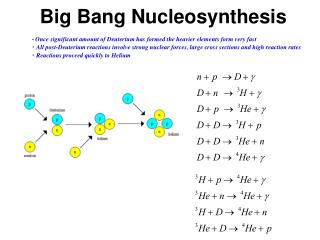
Nucleosynthesis in Supernovae explosions Nucleosynthesis in final stages of a star before Supernovae (or white dwarf) Fe, Ni, Most stable nuclei Unstable to a-decay, fission Heats the earth Fusion of protons to 4He Drives the sun E=mc2
Nuclear Fusion Reactions • Energy release in fusing 6 p + 6 n 12C. • 6 (p) + 6 (n) 0 = 92,16 MeV released • Fractional energy release ≈ 1% • Mass conservation violated at 1% level • Compare to chemical energy • e+p H: Energy Release = (13 eV)/(938 MeV) ≈ 10. • Mass is conserved (parts per billion) in chemical reactions • Energy Release in pp chain • HHHH 4He • 4 (p)- (4He) = 4(8.071 MeV)(2.424 MeV)= 30 MeV