Acid-Base Chemistry
1.07k likes | 1.38k Views
Acid-Base Chemistry. Assignment #7. Acids and Bases - 3 Definitions. Arrhenius Bronsted-Lowry Lewis. Arrhenius Definition. Acid = proton donor HA = H + + A - Base = hydroxide donor BOH = B + + OH - Dilemma: NH 3. Bronsted-Lowry Definition. Acid = proton donor

Acid-Base Chemistry
E N D
Presentation Transcript
Acid-Base Chemistry Assignment #7
Acids and Bases - 3 Definitions • Arrhenius • Bronsted-Lowry • Lewis
Arrhenius Definition • Acid = proton donorHA = H+ + A- • Base = hydroxide donorBOH = B+ + OH- • Dilemma: NH3
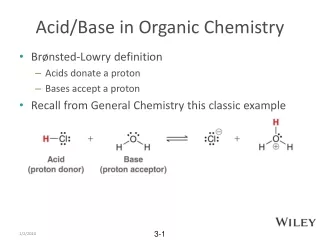
Bronsted-Lowry Definition • Acid = proton donor • Base = proton acceptorNH3 + H+ = NH4+ • Dilemna: ferrocene (organometallics)
Lewis Definition • Acid = electron pair acceptor (electrophile) • Base = electron pair donor (nucleophile)
Acid and Base Strength • Strong acids and Bases completely dissociate (ionize) in aqueous solution • EX:HClaq-> H+aq + Cl-aq • Weak acids and Bases incompletely dissociate in aqueous solution • EX:HC2H3O2aq= H+aq + C2H3O2-aq
Not All Mineral Acids are Strong! • HCNHCN = H+ + CN- Ka = 2.1 x 10-9 • HFHF = H+ + F- Ka = 6 x 10-4
Polyprotic Acids • Protons are always lost one at a time! • acids produced by proton loss from polyprotic acids are weak acids, characterized by a unique Ka value • H2SO4 = HSO4- + H+ Ka1 not measurableHSO4- = SO42- + H+ Ka2 = 1.1 x 10-2
Strong Acids • Most mineral acids • HCl • H2SO4 • HNO3 • HClO4 • Not: HF, H3PO4
Weak Acids • Organic acids (need C and usually have COOH)EXAMPLES:formic acidacetic acidpropionic acid
Strong Bases • Inorganic hydroxides containing metals from families IA or IIA • Note: names of these families: Alkali metals, alkaline earth metals
Weak Bases • NH3, organic amines, and hydroxides other than group Ia or IIa hydroxides • organic amines contain amino group:R-NH2 + H+ = R-NH3+
Practice • Identify the acid/base nature of the following compounds: • Hydroxyl amine • Calcium hydroxide • Carbon dioxide • Pthalic acid • Hydrogen sulfide • pyridine
Conjugates • Acids and Bases exist in a conjugate relationship:HA = H+ + A-acid baseBOH = B+ + OH-base acid
Example: • NH4OH = NH4+ + OH- base acid • HC2H3O2 = H+ + C2H3O2- acid base
Problem • Identify the conjugate acid-base pairs for each of the following compounds: • Ammonium hydroxide • Diethylamine • Iodic acid • Formic acid • HPO4-
Amphoterism • Some compounds can function as both acids or bases depending on the situatione.g., H2O • HC2H3O2 + H2O = H 3O+ + C2H3O2-acid base acid base • NH3 + H2O = NH4+ + OH-base acid acid base
Dissociation Constants for Weak Acids and Bases • Recall for HA: • Ka = [H+][A-] [HA] • The bigger Ka, the _____ the [H+] and the _____ the [HA]
Question • What is the comparatively strongest weak acid on Table A? • ANS: iodic acid, Ka = 0.18 • What is the comparatively strongest weak base on Table B? • ANS: diethylamine and piperidine are equally strong, Kb = 0.0013
Conjugate Acid-Base Strength • For HA = H+ + A- • Recall,Ka = [H+][A-] [HA] • HA is the conjugate _____ and A- is its conjugate ____ • HA is a ____ ____ • A- is a ____ ____ • If HA is a relatively strong weak acid, then A- is a comparatively ____ ____ base
Problem • HSO4- isa) the conjugate acid of SO4-2b) a strong acidc) the conjugate base of H2SO4d) a strong basee) the conjugate acid of H2SO4
Problem: • Which of the following is/are amphoteric:a) H2PO4-b) C2H3O2-c) CH3CH2NH2d) HCO3-e) CH3CH(NH2)COOH
Conjugate Acid-Base Strength • The stronger the conjugate acid is, the weaker the conjugate base is and vice versa HA A-
Salts • There are four kinds of salts: • salts of weak acidsexample: sodium acetate • salts of weak basesexample: ammonium chloride • salts of strong acids and strong basesexample: sodium chloride • salts of weak acids and weak basesexample: ammonium acetate
Identification of Salts • Salts hydrolyze in water: Salt + water acid + base EX: NaCl + HOH Na+ + OH- + H+ + Cl-
Identification of Salts • Salts are obtained by reacting acids and bases: Acid + Base = Salt + water • Note: this is just the reverse of the hydolysis reaction: Salt + water = Acid + Base
Identification of Salts • So, salts are classified based on their parent acids • Their parents are the acids and bases used to form them • EX: NaCl NaOH + HCl Na+ + Cl- + H2O So, parents are strong acid and strong base and NaCl is the salt of a strong acid and strong base
Examples: • NaC2H3O2 – salt of ________________ • NH4Cl – salt of ___________________ • NH4COOH – salt of _______________ • LiF – salt of _____________________
Homework Problem #1: • Classify each of the following as a strong or weak acid, base, or salt (identify parents):CsOH AgOH sodium salicylate H2PO4-HClO4 H2CO3ferric hydroxide oxalic acidNH4C2H3O2trimethylamine
The Autoionization of Water • HOH(l) + HOH(l) = H3O+ + OH-hydronium ion • This is an equilibrium process and is characterized by an equilibrium constant, Kw:Kw = [H3O+][OH-] = 10-14 at 250C
Kw DOES vary with Temperature lower higher
The Relationship between [H+] and [OH-] • Kw = [H+][OH-] = 10-14Useful Equivalent forms: • [H+] = 10-14/[OH-] • [OH-] = 10-14/[H+]
The pH Scale • pH = -log [H+] ; no units • [H+] = antilog[-pH] • pH of pure water = 7
A Brief Review of LOG Math • Taking a logarithm corresponds to answering the question:To what power do I raise 10 in order to represent the number of interest?log 100 => 10? = 100 • NOTE: LOG is not same thing as LNnatural log is based on e?= number
A Brief Review of LOG Math • log (ab) = log a + log b • log (a/b) = log a - log b • log ab = b log a
The pOH scale • pOH = -log [OH-] ; no units • [OH-] = antilog[-pOH] • What is the pOH in pure water? • ANS: pOH = -log (10-7) = 7
How is pH related to pOH? • recall: Kw = [H3O+][OH-] = 10-14 at 250C • Derivation • call pKw = -logKw = 14 at 250C • then:pKw = pH + pOH = 14
Aspirin and Ibuprofen • Clean and Clear Sensitive Skin Deep Cleaning Astringent • active ingredient 0.5% salicylic acid • Advil • active ingredient ibuprofen
Orange Juice • Tropicana Pure Premium with Calcium • active ingredients: calcium hydroxide, malic acid and citric acid
Toothpaste • Aquafresh Whitening Toothpaste • active ingredient: sodium fluoride • Colgate Maximum Cavity Protection Fluoride Toothpaste • active ingredient: 0.76% sodium monofluorophosphate
The Relationship between pH and pOH acidic neutral basic
Acidic Solution • high [H+] concentration • low pH valuevalue below 7 • low [OH-] concentration • high pOH valuevalue greater than 7
Basic Solution • high [OH-] concentration • low pOH valuevalue less than 7 • low [H+] concentration • high pH valuevalue greater than 7
pKa and pKb • pKa≡ - log Ka • pKb ≡ - log Kb • pKa * pKb = pKw
Conjugate Acid-Base Strength • The stronger the conjugate acid is, the weaker the conjugate base is and vice versa • Ka * Kb = 10-14 • pKa + pKb = 14 HA A-
pKa , pKb, and Weak Acid/ Base Strength • The lower the pKa the ______ the weak acid • The higher the pKa, the ______ the weak acid • The lower the pKa of a weak acid, the ______ the pKb of its conjugate weak base and the _____ its conjugate base HA A- pKa * pKb = 14
Question • Which is the comparatively stronger weak acid ammonium or pyridinium?
Strong Acids and Strong Bases • HA → H+ + A- • BOH → B+ + OH- • Strong acids and strong bases completely dissociate in water so their concentration gives us the [H+] in solution directly sea slugs secrete H2SO4 sea squirts squirt HNO3 Photographs from Atkins, P.W. Molecules; W.H. Freeman: New York, 1987.