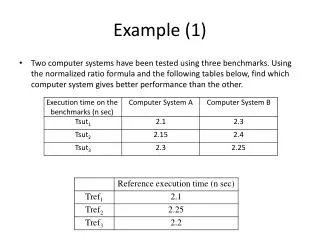
EXAMPLE 11. 1
EXAMPLE 11. 1. Writing Equations. Solution. From Table 11.2, we see that hematite is Fe 2 O 3 . From Chapter 7, we know that hydrochloric acid is HCl. We are told in the example that the products are FeCl 3 and H 2 O so we now have enough information to write an unbalanced equation.

EXAMPLE 11. 1
E N D
Presentation Transcript
EXAMPLE 11. 1 Writing Equations Solution From Table 11.2, we see that hematite is Fe2O3. From Chapter 7, we know that hydrochloric acid is HCl. We are told in the example that the products are FeCl3 and H2O so we now have enough information to write an unbalanced equation. Fe2O3 + HCl FeCl3 + H2O (not balanced) To balance the equation, we see that there are two Fe atoms on the left and only one on the right, so we add a coefficient of 2 to FeCl3. Fe2O3 + HCl 2 FeCl3 + H2O (not balanced) Now there are six Cl atoms on the right, so a coefficient of 6 is needed for HCl. Fe2O3 + 6 HCl 2 FeCl3 + H2O (not balanced) Finally, there are three O atoms and six H atoms on the left, so a 3 in front of H2O will complete the task. Fe2O3 + 6 HCl 2 FeCl3 + H2O (balanced) Exercise 11.1B What mass in grams of HF is needed to dissolve 12 g of quartz? Exercise 11.1A Quartz dissolves in hydrofluoric acid to give silicon tetrafluoride and water. Write and balance an equation for this process. You are told by a geoscientist that hematite (Table 11.2) can be dissolved in hydrochloric acid to form FeCl3 and water. Write a balanced equation for the process.
EXAMPLE 11.2 Energy Calculations Solution We haven’t yet discussed energy in detail, but there is enough information in the paragraph to permit us to do the calculation using dimensional analysis. The paragraph notes that 6300 kJ of energy is needed to make a single disposable bottle, that 8300 kJ is needed to make a single returnable bottle, and that the returnable is used an average of 12.5 times. That means that one returnable is the equivalent of 12.5 disposables. We begin by calculating the number of disposables equivalent to 10,000 returnables. 10,000 returnables x Now we can calculate the energy cost of the disposables, using the factor 6300 kJ = 1 disposable. 125,000 disposables x Then we can calculate the energy cost of the returnables in a similar way. 10,000 returnables x Notice that we can do this calculation without knowing just what a joule is! Exercise 11.2 12.5 disposables = 125,000 disposables When one mole (16.0 g) of methane is burned in air, 890 kJ of energy is produced. What mass of methane is equivalent to the energy difference between the returnables and the disposables in Example 11.2? 1 returnable 6300 kJ = 7.88 x 108 kJ 1 disposable 8300 kJ = 8.3 x 107 kJ 1 returnable Based on the data in the previous paragraph, calculate the quantity of energy needed to produce 10,000 returnable bottles, and compare it to the energy needed to produce the equivalent number of disposable bottles.