Proteins
Proteins are essential multipurpose molecules that perform a vast array of functions in living organisms. They are composed of amino acids, with 20 different types forming polypeptide chains that fold into intricate three-dimensional shapes. Proteins act as enzymes, structural components, transporters, receptors, and hormones. Their structure is critical to their function, influencing everything from catalyzing biochemical reactions to supporting cellular integrity. Understanding how proteins are built and how their structure impacts functionality is vital in biology, medicine, and biochemistry.

Proteins
E N D
Presentation Transcript
Proteins Multipurposemolecules
Proteins • Most structurally & functionally diverse group of biomolecules • Function: • involved in almost everything • enzymes (pepsin, polymerase, etc.) • structure (keratin, collagen) • carriers & transport (membrane channels) • receptors & binding (defense: antibodies) • contraction (actin & myosin) • signaling (hormones: insulin) • storage (bean seed proteins)
Proteins • Structure: • monomer =amino acids • 20 different amino acids • polymer =polypeptide • protein can be one or more polypeptide chains folded & bonded together • large & complex molecules • complex 3-D shape hemoglobin growthhormones Rubisco
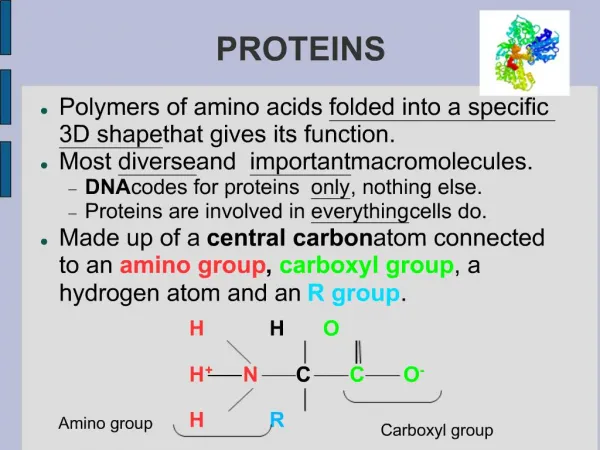
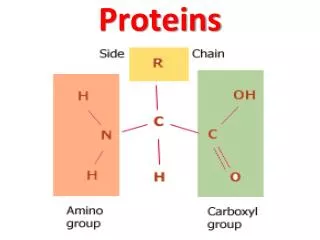
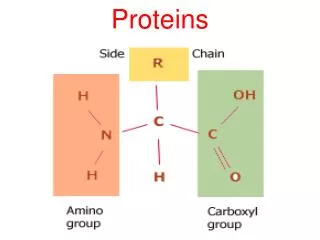
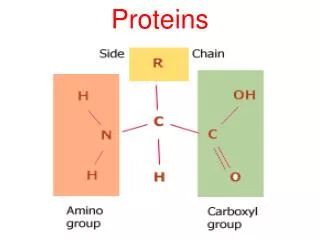
H O | —C— | H || C—OH —N— H Amino acids • Structure: • central carbon • amino group • carboxyl group (acid) • R group (side chain) • variable group • confers unique chemical properties of the amino acid R
nonpolar & hydrophobic Nonpolar amino acids Why are these nonpolar & hydrophobic?
polar or charged & hydrophilic Polar amino acids Why are these polar & hydrophillic?
Sulfur containing amino acids • Formdisulfide bridges • cross links betweens sulfurs in amino acids H-S – S-H You wonderedwhy permssmelled like rotten eggs?
peptidebond Building proteins • Peptide bonds • linking NH2 of one amino acid to COOH of another • C–N bond dehydration synthesis
Protein models • Protein structure visualized by • X-ray crystallography • extrapolating from amino acid sequence • computer modelling lysozyme
Building proteins • Polypeptide chains • N-terminus = NH2 end • C-terminus = COOH end • repeated sequence (N-C-C) is the polypeptide backbone • can only grow in one direction
hemoglobin collagen Protein structure & function • Function depends on structure • 3-D structure • twisted, folded, coiled into unique shape pepsin
Primary (1°) structure • Order of amino acids in chain • amino acid sequence determined by gene (DNA) • slight change in amino acid sequence can affect protein’s structure & it’s function • even just one amino acid change can make all the difference! lysozyme: enzyme in tears & mucus that kills bacteria
Secondary (2°) structure • “Local folding” • folding along short sections of polypeptide • interaction between adjacent amino acids • H bonds between R groups • -helix • -pleated sheet
“Let’s go to the video tape!” (play movie here) Secondary (2°) structure
Tertiary (3°) structure • “Whole molecule folding” • determined by interactions between R groups • hydrophobic interactions • effect of water in cell • anchored by disulfide bridges(H & ionic bonds)
Quaternary (4°) structure • More than one polypeptide chainjoined together • only then is it a functional protein • hydrophobic interactions collagen = skin & tendons hemoglobin
Chaperonin proteins • Guide protein folding • provide shelter for folding polypeptides • keep the new protein segregated from cytoplasmic influences
Denature a protein • Unfolding a protein • disrupt 3° structure • pH salt temperature • unravels or denatures protein • disrupts H bonds, ionic bonds & disulfide bridges • destroys functionality • Some proteins can return to their functional shape after denaturation, many cannot In Biology,size doesn’t matter, SHAPE matters!
multiplepolypeptides hydrophobic interactions aa sequence peptide bonds determinedby DNA R groups H bonds Protein structure (review) R groups hydrophobic interactions, disulfide bridges 3° 1° 2° 4°
Let’s build some Proteins!