Water
200 likes | 327 Views
Water. Importance of Water. More than 70% of our total body weight is water Necessary for photosynthesis H becomes incorporated into organic compounds Oxygen released for us to breathe Solvent for most biochemical reactions Important reactant/product. Water is a polar molecule.

Water
E N D
Presentation Transcript
Importance of Water • More than 70% of our total body weight is water • Necessary for photosynthesis • H becomes incorporated into organic compounds • Oxygen released for us to breathe • Solvent for most biochemical reactions • Important reactant/product
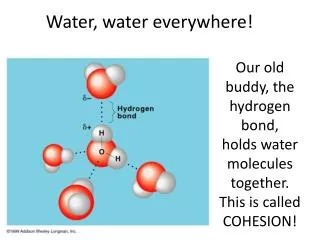
Water is a polar molecule • Due to differences in electronegativities, water is slightly charged at its poles • Oxygen takes on a slight – charge • Hydrogens take on a slight + charge • http://programs.northlandcollege.edu/biology/Biology1111/animations/hydrogenbonds.html
Hydrogen Bonding • Water can form H-bonds with up to 4 neighboring water molecules
It is water’s polarity that gives it many of its unique properties • Less dense as a solid than a liquid • Universal solvent • Adhesion and cohesion • Capillary action • Surface tension • High heat of vaporization • High specific heat
Water is the universal solvent • Water can dissolve many hydrophilic substances • Ionic compounds • Other polar compounds • Form “spheres of hydration” • http://www.mhhe.com/physsci/chemistry/essentialchemistry/flash/molvie1.swf
Some substances do not dissolve readily in water • Hydrophobic – “water-fearing” • Non-polar substances like lipids
Cohesion • Water molecules have a strong tendency to stick to one another • Cohesion allows water to have a high degree of surface tension • Any force is transmitted to the column of water as a whole
Adhesion • Ability of water to stick to other substances, esp. charged atoms or molecules • Together, cohesion and adhesion allow for capillary action
How Unique is Water?? • Water is one of only 3 naturally occurring inorganic liquids (mercury and ammonia) • Only chemical compound that exists in all 3 states—solid, liquid, and gas • Extremely large liquid range (0oC - 100oC) • Expands, becomes less dense as a solid
Water’s 3 states differ in the degree of H-bonding http://mutuslab.cs.uwindsor.ca/schurko/animations/waterphases/status_water.htm
Liquid water has • H-bonds that form and break constantly • http://www.stolaf.edu/people/giannini/flashanimat/water/water.swf • Allows water to have a high specific heat • Amount of energy required to raise temp of 1 g 1o C • 1 cal/1 g
Water Vapor • As water moves from liquid to gaseous state, H-bonds are broken, allowing water molecules to escape • Water has a high heat of vaporization: • It takes 540 cal for 1 g of water to move from liquid to gaseous state • Allows for evaporative cooling • As fast-moving liquid water molecules escape as vapor, they take their heat energy with them
Ice • Solid water is less dense than liquid water, allowing it to float
Acids and Bases • Water molecules have slight tendency to ionize: H2O < -- > H+ + OH- • The H+ then joins another water molecule resulting in H3O+ (hydronium) • The pH scale is a measure of hydronium concentration expressed in moles/liter
pH scale • http://www.johnkyrk.com/H2O.html • pH = -log10[H+] • Acids are proton donors • Increase the # of H+ ions • Bases are proton acceptors
Acid + Base Salt • Acid = H+ + anion • Base = OH- + cation • H+ joins with OH- to get H2O • Anion can combine with cation to make a salt • Ex. HCl + NaOH H2O + NaCl
Buffers • Substance(s) that resist pH changes when an acid or base is added • Usu. weak acid or base, do not completely ionize • Example: Blood in Vertebrates
CO2 + H20 < -- > H2CO3< -- > H+ + HCO3- • Will stay at dynamic equilibrium unless stressed • If add excess H+ system shifts left and forms carbonic acid • If add OH- they combine with H+ forming water, system shifts right • http://www.tvdsb.on.ca/westmin/science/sbioac/biochem/buffer.htm • http://www.mhhe.com/physsci/chemistry/essentialchemistry/flash/buffer12.swf