Partial Molar Variables, Chemical Potential, Fugacities, Activities, and Standard States
I 2 (aq). I 2 (CCl 4 ). Partial Molar Variables, Chemical Potential, Fugacities, Activities, and Standard States

Partial Molar Variables, Chemical Potential, Fugacities, Activities, and Standard States
E N D
Presentation Transcript
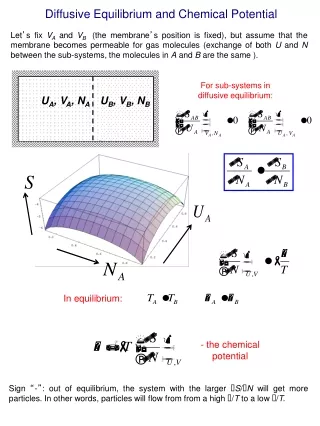
I2 (aq) I2 (CCl4) Partial Molar Variables, Chemical Potential, Fugacities, Activities, and Standard States Partial molar thermodynamic variables with respect to species i describe the differential change in a thermodynamic variable as the number of moles of species i is differentially changed while holding the temperature, pressure and number of moles of all other species constant. For example the partial molar volume of species i is: Vi ( V / ni)T, P,nj The chemical potential of species iis defined as species i’s partial molar Gibbs’ free energy: ui Gi ( G / ni)T, P,nj If molecular iodine dissolved in water, I2 (aq), is in equilibrium with molecular iodine dissolved in CCl4, I2 (CCl4), what is the relation between the chemical potentials of molecular iodine in each of these immiscible phases? 38.1
As an example of using the definition of chemical potential, let’s calculate how the absolute molar Gibbs’ free energy of a single ideal gas varies isothermally with pressure. We Begin with: dG = V dP dT = 0 and separate variables and integrate from the standard pressure of 1 bar: GoTGT,P dG = 1 bar P (n R T / P) dP After dividing the result of the integration by the number of moles, n, we get: GT,P = GoT + R T ln (P / 1 bar) Noting that the partial molar Gibbs’ free energy of a single species is just its molar Gibbs’ free energy, we can write this result in terms of chemical potentials: uT,P = uoT + R T ln (P / 1 bar) This result is easily extended to give the chemical potential of an ideal gas in a mixture of ideal gases as: ui,T ,P = uoi,T + R T ln (Pi / 1 bar) 38.2
direction of He diffusion uHe, outside< uoHe PHe@ 1 bar uHe, inside@uoHe When He filled balloons go flat, you are observing the result of the small He atoms diffusing slowly through the porous membrane of a rubber balloon: In order to inflate the balloon when the outside pressure is 1 bar, the pressure of the He inside the balloon must be slightly greater than 1 bar: uHe, inside = uoHe + R T ln (PHe@ 1 bar / 1 bar) @ uoHe In a normal atmosphere the pressure of He outside the balloon is approximately zero, thus the chemical potential of the He inside the balloon is substantially greater than the chemical potential of He outside the balloon: (uHe, inside@ uoHe ) > (uHe, outside< uoHe ) Note that the He is observed to diffuse or move from a region of high chemical to a region of low chemical potential. Balls rolling down hills move from positions of high potential energy to positions of low potential energy. Do you see an analogy with the movement of chemical species between regions of differing chemical potential? 38.3
In order to preserve the convenient form of the expression we have just derived for ideal gases and yet have an expression which is valid for real gases we define the fugacity, f, such that the expression: ui, T, P = uoi, T + R T ln (fi / f o = 1 bar) holds exactly for real gases at all pressures. f o = 1 baris the standard state fugacity. The fugacity can be viewed as kind of an equivalent pressure, has units of pressure, and as we shall see in a moment can be calculated from the pressure given an equation of state or PVT data. For an ideal gas how are the fugacity and the pressure of the gas related? Since solids and liquids have finite, if sometimes immeasureably small, vapor pressures, we can extend the application of the above expression to solids and liquids, as well as gases. Here it is convenient to define theactivity of species ias: ai fi / f o This definition now allows us to express the chemical potential of any solid, liquid, or gaseous species as: ui, T, P = uoi, T + R T ln ai When species i is in its standard state, what is its activity? 38.4
When a substance is in its standard state its activity is equal to unity; ai = fi / f o= 1 its fugacity is equal to its standard state fugacity; fi = f o= 1 bar and its chemical potential is equal to its standard state chemical potential: ui,T, P = uoi,T Since by definition the fugacity of an ideal gas is equal to its pressure at all pressures: f ideal gas = P ideal gas an ideal gas is in its standard state when its pressure is 1 bar at some specified temperature. 38.5
ideal gas fi = Pi hypothetical standard state fugacity real gas f = 1 bar not the standard state P = 1 bar pressure A real gas is in its standard state when its fugacity is equal to 1 bar and it is behaving as if it were an ideal gas at some specified temperature (the diagram shown below is exaggerated to make this point): In the limit of zero pressure real gases behave more and more like ideal gases and the fugacities of real gases approach their partial pressures: lim fi --> PiPi --> 0 At room temperature what approximately is the fugacity of a real gas when its pressure is 1 bar? What would its activity be in this case? The standard state of a pure liquid or solid is the pure liquid or solid at 1 bar of pressure and some specified temperature. Since the molar volumes of solids and liquids are generally small and relatively insensitive to pressure, the activities of solids and liquids at pressures that are not too far removed from 1 bar remain close to unity. 38.6
As an example of the insensitivity of the activity of condensed phases to variations in pressure, we will calculate the activity of pure liquid water at 25.0 oC while under the equilibrium pressure of its vapor of 23.76 mm Hg? We can start by rearranging: uH2O (l), T = uoH2O (l), T + R T ln aH2O (l) to give: aH2O (l) = e + (u - uo) / R T Recalling that the isothermal pressure dependence of the Gibbs’ free energy is given by: dG = V dP and that the chemical potentials of pure substances are just their molar Gibbs’ free energies, we can express the difference in chemical potentials as: uH2O (l), T - uoH2O (l), T = GH2O (l), T - GoH2O (l), T = 1 bar P VH2O (l) dP @ VH2O (l) (P - 1 bar) Substituting this result into the expression for the activity of liquid water: aH2O (l) = e + V H2O (l) (P - 1 bar) / R T = e + (18.07 mL /mol) (0.03168 bar - 1 bar) / [(83.14 mL bar / mol K) (298.2 K)] = e - 0.0007058 = 0.99929 we see that the activity of liquid water at 23.76 mm Hg (or 0.013168 bar) is still close to unity. 38.7
To see how to calculate the fugacity of a single real gas from its pressure we begin by differentiating: ln (f / P) with respect to pressure while holding the temperature constant to get: ( ln (f / P) / P)T = ( ln f / P)T - 1 / P Now using: u = uo + R T ln a= uo + R T ln (f / f o ) we can write: ( ln (f / P) / P)T = ( [(u - uo) / (R T) + ln f o] / P)T - 1 / P = (1 / R T) ( u / P)T - 1 / P Can you justify this last equality? Now using the fact that the chemical potential and molar Gibbs’ free energy of a single species are one and the same and that the partial derivative of the molar Gibbs’ free energy with respect to pressure, taken while holding the temperature constant, is equal to the molar volume: ( u / P)T = ( G / P)T = V / n we obtain the equation: ( ln (f / P) / P)T = V / (n R T) - 1 / P 38.8
The variables in this equation can be separated and integrated from zero pressure to P: 0 ln (f / P)d ln (f / P)= 0 P[V / (n R T) - 1 / P] dP Why is the lower limit on the left-hand integral zero when P = 0? Integrating the left side of this equation gives anequation for calculating the fugacity of a real gas from its pressure: ln ( f ) = ln P + 0 P [V / (n R T) - 1 / P]dP What does this equation predict for the fugacity of an ideal gas at 20 bar? The virial equation of statecan be written as an expansion in pressure: P V = n R T [1 + B P + C P2 + … ] where the virial coefficients are only functions of temperature and the nature of the gas. Derive an equation that would allow you to calculate the fugacity of a gas following this virial equation of state from the gas pressure. 38.9
Bonus Problem Fugacity of a Van der Waals Gas This problem is worth 20 points and is due 1 week from the date that it is assigned. The van der waals equation is a real gas equation of state: P = n R T / (V - n b) - a n2 / V2 In this equation the van der Waals constant, "b", is a measure of the finite, but small, actual molar volume of the molecules while, "a", accounts for the intermolecular attractive interactions between molecules. For CO2 the van der Waals constants are: a = 3.658 L2 bar / mole2 b = 0.0429 L / mole Calculate the fugacity of 1.000 mole of CO2 in bar at a temperature of 350.0 K and a pressure to be assigned in class between 15.00 bar and 25.00 bar. You will have to write a program or use a spreadsheet to: 1. iteratively solve the van der Waals equation for volume, using Newton's method, at each of, say 100, pressures between zero pressure and the pressure of interest; 2. then numerically integrate: 0 P [ V / (R T) - 1 / P ] dP using Simpson's rule. To test your program you can use the result that when P = 25.00 bar, the fugacity of CO2 is 23.26 bar. 38.10