Spatial Learning and Memory in the Sand Maze: WIN-2 Effects on State-Dependent Retention in Rats
E N D
Presentation Transcript
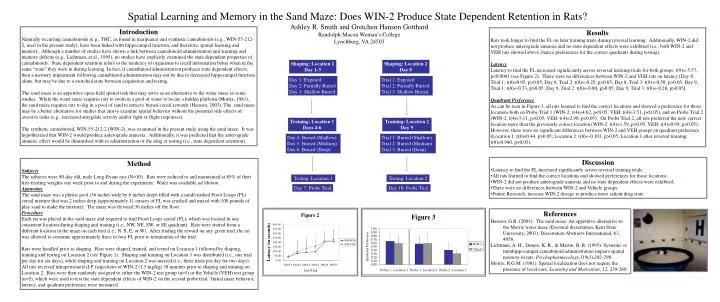
Spatial Learning and Memory in the Sand Maze: Does WIN-2 Produce State Dependent Retention in Rats? Ashley R. Smith and Gretchen Hanson Gotthard Randolph-Macon Woman’s College Lynchburg, VA 24503 Introduction Naturally occurring cannabinoids (e.g., THC, as found in marijuana) and synthetic cannabinoids (e.g., WIN-55-212-2, used in the present study), have been linked with hippocampal function, and therefore, spatial learning and memory. Although a number of studies have shown a link between cannabinoid administration and learning and memory deficits (e.g., Lichtman, et al., 1995), no studies have explicitly examined the state dependent properties of cannabinoids. State dependent retention refers to the tendency of organisms to recall information better when in the same “state” they were in during learning. In fact, if cannabinoid administration produces state dependent effects, then a memory impairment following cannabinoid administration may not be due to decreased hippocampal function alone, but may be due to a switched state between acquisition and testing. The sand maze is an appetitive open-field spatial task that may serve as an alternative to the water maze in some studies. While the water maze requires rats to swim in a pool of water to locate a hidden platform (Morris, 1981), the sand maze requires rats to dig in a pool of sand to retrieve buried cereal rewards (Hanson, 2003). The sand maze may be a better alternative for studies that aim to examine spatial behavior without the potential side effects of aversive tasks (e.g., increased amygdala activity and/or fight or flight responses). The synthetic cannabinoid, WIN-55-212-2 (WIN-2), was examined in the present study using the sand maze. It was hypothesized that WIN-2 would produce anterograde amnesia. Additionally, it was predicted that the anterograde amnesic effect would be diminished with re-administration of the drug at testing (i.e., state dependent retention). Results Rats took longer to find the FL on later training trials during reversal learning. Additionally, WIN-2 did not produce anterograde amnesia and no state dependent effects were exhibited (i.e., both WIN-2 and VEH rats showed above chance preferences for the correct quadrants during testing). Latency Latency to find the FL increased significantly across reversal learning trials for both groups, t(9)=-5.57, p<0.0001 (see Figure 2). There were no differences between WIN-2 and VEH rats on latency [Day 8, Trial 1: t(8)=0.05, p>0.05; Day 8, Trial 2: t(8)=-0.25, p>0.05; Day 8, Trial 3: t(8)=-0.50, p>0.05; Day 9, Trial 1: t(8)=-0.73, p>0.05; Day 9, Trial 2: t(8)=-0.80, p>0.05; Day 9, Trial 3: t(8)=-0.28, p>0.05]. Quadrant Preference As can be seen in Figure 3, all rats learned to find the correct locations and showed a preference for those locations both on Probe Trial 1 (WIN-2: t(4)=4.62, p<0.05; VEH: t(4)=3.51, p<0.05), and on Probe Trial 2 (WIN-2: t(4)=3.11, p<0.05; VEH: t(4)=2.90, p<0.05). On Probe Trial 2, all rats preferred the new correct location more than the previously correct location (WIN-2: t(4)=1.59, p>0.05; VEH: t(4)=0.99, p>0.05). However, there were no significant differences between WIN-2 and VEH groups on quadrant preference (Location 1: t(8)=0.44, p>0.05; Location 2: t(8)=-0.103, p>0.05; Location 1 after reversal training: t(8)=0.940, p>0.05). 180˚ Shaping: Location 1 Day 1-3 Shaping: Location 2 Day 8 Day 1: Exposed Day 2: Partially Buried Day 3: Shallow Buried Trial 1: Exposed Trial 2: Partially Buried Trial 3: Shallow Buried Training: Location 1 Days 4-6 Training: Location 2 Day 9 Day 4: Buried (Shallow) Day 5: Buried (Medium) Day 6: Buried (Deep) Trial 1: Buried (Shallow) Trial 2: Buried (Medium) Trial 3: Buried (Deep) • Discussion • Latency to find the FL increased significantly across reversal training trials. • All rats learned to find the correct locations and showed preferences for those locations. • WIN-2 did not produce anterograde amnesia and no state dependent effects were exhibited. • There were no differences between WIN-2 and Vehicle groups. • Future Research: increase WIN-2 dosage to produce more salient drug state. Method Subjects The subjects were 90-day old, male Long-Evans rats (N=10). Rats were reduced to and maintained at 85% of their free-feeding weights one week prior to and during the experiment. Water was available ad libitum. Apparatus The sand maze was a plastic pool (36 inches wide by 6 inches deep) filled with a sand/crushed Froot Loops (FL) cereal mixture that was 2 inches deep (approximately 11 ounces of FL was crushed and mixed with 100 pounds of play sand to make the mixture). The maze was elevated 36 inches off the floor. Procedure Each rat was placed in the sand maze and required to find Froot Loops cereal (FL), which was located in one consistent location during shaping and training (i.e., NW, NE, SW, or SE quadrant). Rats were started from a different location in the maze on each trial (i.e., N, S, E, or W). After finding the reward on any given trial, the rat was allowed to consume approximately three to four FL prior to termination of the trial. Rats were handled prior to shaping. Rats were shaped, trained, and tested on Location 1 followed by shaping, training and testing on Location 2 (see Figure 1). Shaping and training on Location 1 was distributed (i.e., one trial per day for six days), while shaping and training on Location 2 was massed (i.e., three trials per day for two days). All rats received intraperitoneal (I.P.) injections of WIN-2 (1.5 mg/kg) 30 minutes prior to shaping and training on Location 2. Rats were then randomly assigned to either the WIN-2 test group (n=5) or the Vehicle (VEH) test group (n=5), which were used to test the state dependent effects of WIN-2 on the second probe trial. Initial maze behavior, latency, and quadrant preference were measured. Testing: Location 1 Testing: Location 2 Day 7: Probe Trial Day 10: Probe Trial References Hanson, G.R. (2003). The sand maze: An appetitive alternative to the Morris water maze (Doctoral dissertation, Kent State University, 2003). Dissertation Abstracts International, 63, 4958. Lichtman, A. H., Dimen, K. R., & Martin, B. R. (1995). Systemic or intrahippocampal cannabinoid administration impairs spatial memory in rats. Psychopharmacology,119(3),282-290. Morris, R.G.M. (1981). Spatial localization does not require the presence of local cues. Learning and Motivation, 12, 239-260.