Comprehensive Study Notes on Solutions: Definitions, Properties, and Calculations
This study guide covers essential concepts about solutions, including terminology such as solute, solvent, saturated, and unsaturated solutions. It explains the polarity of molecules, solubility rules, and factors affecting solubility like temperature and pressure. The guide also outlines molarity and molality calculations, providing formulas for dilutions and predicting ionic compound solubility. Finally, it introduces colligative properties, explaining their relevance in solution chemistry. A must-read for exam preparation in chemistry!

Comprehensive Study Notes on Solutions: Definitions, Properties, and Calculations
E N D
Presentation Transcript
Solutions Unit Final Exam Study Notes
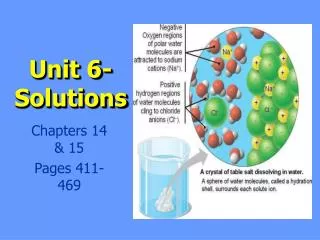
What is a solution? • Solution Terminology: Solute, solvent, saturated, unsaturated, supersaturated, aqueous, homogeneous, heterogeneous, soluble, insoluble, miscible (alcohol and water), immiscible (water and oil), dilute, concentrated Polar, nonpolar (molecules based on VESPR and electronegativities)
Polar molecules dissolve other polar molecules and ionic compounds • Nonpolar molecules dissolve other nonpolar molecules. • Alcohols will dissolve in both types of solvents but will not dissolve ionic solids. • A saturated solution is an equilibrium system. solute (s, l, g) solute (aq) Temperature affects solubility as well as pressure if the solute is a gas. Increasing the temperature will increase the solubility of solids and liquids but decrease the solubility of gases (exothermic). The solubility of gases will increase with increasing pressure. Stirring as well as increasing the particle size (crushing) will also affect the rate of solubility.
Solubility Curves • Tell us what mass of solute will dissolve in 100 g (mL) of water over a range og temperatures. • For most substances, the solubility will increase as temperature increases. • The line on the graph indicates the concentration of the saturated solution.
Molarity and Molality Calculations • Molarity M = moles solute (mol) liters of solution (L) • This is the concentration [ ] • Molarity will allow us to calculate the grams of the substance needed based on molar mass of the substance • Dilutions C1V1 = C2V2 C = [ ] • Molality m= number of moles (n) kilogram of solvent (kg)
Solubility Rules and Table • Solubility tables will allow us to predict whether a certain ionic compound will dissolve in water. • This determines whether a precipitate will form and from that a net ionic equation can be written.
3 CuSO4(aq) + 2Na3PO4 (aq) Cu3(PO4)2(s) + 3Na2SO4(aq) • Ionic Equation: 3Cu2+(aq) + 3SO42- (aq) + 6Na+(aq) + 2PO43- (aq) Cu3(PO4)2 (s) + 6Na+(aq) + 3SO4 2-(aq) • Net Ionic Equation: 3Cu2+(aq) + 2PO43- (aq) Cu3(PO4)2 (s) • Remember to remove the spectator ions in the final net ionic equation.
Colligative Properties • Properties of solutions that depend on the number of solute particles. • They include boiling point elevation, freezing point depression, vapour pressure and osmotic pressure. • ΔTf = Kfm or ΔTb = Kbm • Kf and Kb are constants m is molality