Heating the Earth
Heating the Earth. Energy From the Sun. Where We Get Our Energy. The Sun is a major source of energy for Earth. . Forms of Energy. Different forms of energy include heat, light, electrical, mechanical, sound, nuclear, and chemical. . Electromagnetic Energy.

Heating the Earth
E N D
Presentation Transcript
Heating the Earth Energy From the Sun
Where We Get Our Energy • The Sun is a major source of energy for Earth.
Forms of Energy • Different forms of energy include heat, light, electrical, mechanical, sound, nuclear, and chemical.
Electromagnetic Energy • Different forms of electromagnetic energy have different wavelengths. • Examples of electromagnetic energy are: • microwaves • infrared light • visible light • ultraviolet light • X-rays • gamma rays.
Electromagnetic Spectrum • The entire range of radiant energy, including for example, X-ray, visible light, infrared radiation, radio waves, etc. • The full set of waves that can travel through space; includes not only visible light but shorter waves, such as X -rays, and longer waves, such as radio waves
Changing Energy • Energy is transformed in many ways.
Changing Energy • Energy can change from one form into another • In the process some energy is always converted to heat.
Conservation of Energy • Energy cannot be created or destroyed, but only changed from one form into another.
Heat Energy • The energy of a material due to the random motion of its particles (atoms 7 molecules). • Also called thermal energy. • “Heat" is used when energy is transferred from one substance to another.
Heat Energy Explains Phases of Matter • The motion of particles (atoms & molecules) helps to explain: • the phase (states) of matter • changes from one phase to another.
Phases (states) of Matter • Gases have neither a determined shape nor a definite volume. Gases assume the shape and volume of a closed container. • Liquids have a definite volume, but take the shape of a container. • Solids have a definite shape and volume. Particles resist a change in position.
Heat Energy Causes Phase Changes • During a phase change, heat energy is absorbed or released. • Energy is absorbed when: • a solid changes to a liquid (melting) • a liquid changes to a gas (evaporation) • a solid changes directly to a gas (sublimation) • Energy is released when: • a gas changes to a liquid (condensation) • a liquid changes to a solid (freezing) • A gas changes directly to a solid, such a forming frost or snow flakes in clouds (crystallization).
Heat Energy Moves • Heat can be transferred through: • matter by the collision of atoms and/or molecules (conduction) • through space (radiation.) • In a liquid or gas, currents will cause the transfer of heat (convection.)
Heat Energy Moves • Heat moves in predictable ways, flowing from warmer objects to cooler ones, until both reach the same temperature
Heat Moves Through the Atmosphere • The transfer of energy within the atmosphere results in formation of regions of different densities. • These density differences result in motion.
Heat & Density • In fluids (liquids and gases), less dense (cooler) substances can rise and more dense (warmer) substances can sink.
Heat Movement & Density in the Atmosphere • The transfer of heat energy within the atmosphere occurs as a result of radiation, convection, and conduction. • Density differences are the basis for many Earth phenomena, including cloud formation and the formation of atmospheric storms.
Heat Movement & Density in the Atmosphere • Heating of Earth’s surface and atmosphere by the Sun drives convection within the atmosphere and oceans, producing winds and ocean currents.
Uneven Heating • Insolation (Incoming solar radiation) heats Earth’s surface and atmosphere unequally due to variations in: • intensity (caused by variations in angle of incidence which vary with time of day, latitude, and season); • characteristics of the surface materials absorbing the energy (such as color, texture, transparency, states of matter, and specific heat); and • duration which varies with seasons and latitude.
http://ithacasciencezone.com/earthzone/lessons/07meteor/insolation.htmhttp://ithacasciencezone.com/earthzone/lessons/07meteor/insolation.htm
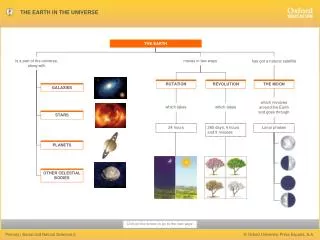
Weather & Seasons • Seasons and weather are NOT the same thing • Seasons are caused by: • Earth’s tilt • Earth’s revolution by around the Sun • Resulting in the Earth getting different intensities of energy from the Sun (heat & light) at different times of the year
Weather & Seasons • Seasonal changes in weather can be explained using concepts of density and heat energy. These changes include: the shifting of global temperature zones, the shifting of planetary wind and ocean current patterns, the occurrence of hurricanes, monsoons, rainy and dry seasons, flooding, severe weather, and ozone depletion.
Big Science Idea: • The atmosphere is an ocean of air that extends hundreds of kilometers about the Earth's surface. • It is divided according to temperature into four layers - the troposphere, the stratosphere, the mesosphere, and the thermosphere. • The lowest layer, the troposphere is where air masses form. • Air masses have different characteristics because the areas over which they form receive different amounts of heat energy from the sun. • The uneven heating of Earth’s surface is the cause of weather.
Let’s Review: http://wps.prenhall.com/esm_lutgens_atmosphere_8/0,6585,263598-,00.html
References • http://www.earth2class.org • http://universityforholisticarts.org/files/QuickSiteImages/QS_sun_background.jpg • http://science.uniserve.edu.au/school/curric/k_6/solar.jpg • http://genesismission.jpl.nasa.gov/science/mod3_SunlightSolarHeat/img1.gif • http://discover.edventures.com/images/termlib/f/fossil_fuels/support.gif • http://www.stanford.edu/group/Urchin/GIFS/spectrum.gif • http://www.projectgreenmachine.org/7F-all.html • http://www.nasa.gov/centers/langley/images/content/114284main_EM_Spectrum500.jpg • http://www.swinburne.edu.au/corporate/hr/ohs/images/radiofrequency.jpg • www12.mawebcenters.com/coltslaboratories/gloss.ivnu
References • www.riverdeep.net/current/2002/01/010702_light_tg.jhtml • www.yorku.ca/eye/spectru.htm • http://sol.sci.uop.edu/~jfalward/physics17/chapter3/energytransformation.jpg • http://www.tburg.k12.ny.us/mcdonald/chain.jpg.gif • http://www.bchydro.com/images/rx_images/community/body2880.gif • http://www.bioedonline.org/slides/slideimgs/talk007__s014_f.gif • http://www.eia.doe.gov/kids/energyfacts/science/images/EnergyTransformations.gif • http://user.gs.rmit.edu.au/caa/global/graphics/insolation.jpg • http://www.boscobel.k12.wi.us/~schnrich/uneven_heating.htm
References • http://www.miramar.sdccd.cc.ca.us/faculty/fgarces/zCourse/Spring05/Ch100_OL/aMy_FileLec/04_LecNotes_Ch100/13_LiquidSolids/1301_LiquidSolid/1301_liquidSolids.htm • http://www.physics.brocku.ca/courses/1p93/images/FG11_16.gif • www.grc.nasa.gov/WWW/K-12/airplane/heat.html • http://www.cloudappreciationsociety.org/3content/news/hollingshead/i000766big.jpg • http://www.anchorflagandflagpole.com/images/PHOTO_windsock_detail.gif • http://www.nmm.ac.uk/upload/img/currents.gif • http://www.mhhe.com/earthsci/geology/mcconnell/earths_climate/gac.htm