Chemical Equilibrium
Chemical Equilibrium. Chapter 13 AP CHEMISTRY. Chemical Equilibrium. The state where the concentrations of all reactants and products remain constant with time. On the molecular level , there is frantic activity. Equilibrium is not static, but is a highly dynamic situation.

Chemical Equilibrium
E N D
Presentation Transcript
Chemical Equilibrium Chapter 13 AP CHEMISTRY
Chemical Equilibrium • The state where the concentrations of all reactants and products remain constant with time. • On the molecular level, there is frantic activity. Equilibrium is not static, but is a highly dynamic situation.
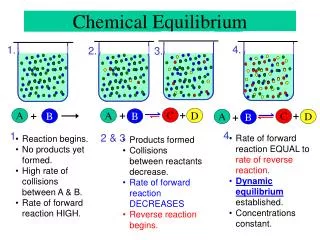
The Concept of Equilibrium Chemical equilibrium occurs when a reaction and its reverse reaction proceed at the same rate.
The Concept of Equilibrium • As the substance warms it begins to decompose: • N2O4(g) 2NO2(g) • A mixture of N2O4 (initially present) and NO2 (initially formed) appears brown. • When enough NO2 is formed, it can react to form N2O4: • 2NO2(g) N2O4(g). • At equilibrium, as much N2O4 reacts to form NO2 as NO2 reacts to re-form N2O4
The Concept of Equilibrium • As a system approaches equilibrium, both the forward and reverse reactions are occurring. • At equilibrium, the forward and reverse reactions are proceeding at the same rate.
A System at Equilibrium Once equilibrium is achieved, the amount of each reactant and product remains constant.
N2O4 (g) 2 NO2 (g) Depicting Equilibrium Since, in a system at equilibrium, both the forward and reverse reactions are being carried out, we write its equation with a double arrow.
[C]l[D]m [A]j[B]k K = Notes on Equilibrium Expressions (EE) • For a given equation: jA + kB ↔lC + mD • K does not include any pure solids or liquids • The expression shows products divided by reactants • Like the rate constant, k, the units of K depend on the experiment being performed • For the reverse reaction K’ = 1/K (reactants and products switch) • Sometimes you will see K written as Kc • Law of mass action
Equilibrium Expression • Write the equilibrium expression for: • 4NH3(g) + 7O2(g) « 4NO2(g) + 6H2O(g) • Complete sample problems #1-4. See sample problems 13.1 and 13.2 in your textbook for more worked examples. Complete sample problems # 1-4.
Significance of Equilibrium Expression • The inherent tendency for a reaction is occur is indicated by the magnitude of K. • A K value much larger than 1 means that at equilibrium the reaction system will consist of mainly product – equilibrium lies to the right • A very small K means that the system at equilibrium will consist mainly of reactants – equilibrium position is far to the left • The size of K and the time required to reach equilibrium are NOT directly related. Complete sample problem #5.
[C]l[D]m [A]j[B]k K = Notes on Equilibrium Expressions (EE) • For a reaction multiplied by an integer, “n”, Knew = (Korig)n • See sample exercise 13.2(c) on page 614. • For a given reaction, K is dependent only on temperature
Heterogeneous Equilibria • . . . are equilibria that involve more than one phase. • CaCO3(s) « CaO(s) + CO2(g) • K = [CO2] • The position of a heterogeneous equilibrium does not depend on the amounts of pure solids or liquids present.
Practice Problem: • Consider the reaction represented by the equation: • Fe3+(aq) + SCN-(aq) FeSCN2+(aq) • In trial #1, you start with 6.00 M Fe3+(aq) and 10.0 M SCN-(aq), and at equilibrium the concentration of FeSCN2+(aq) is 4.00 M. • What is the value of the equilibrium constant for this reaction? Fe3+(aq) + SCN-(aq) FeSCN2+(aq) Initial Change Equilibrium
Practice Problem • Fe3+(aq) + SCN-(aq) FeSCN2+(aq) • Equilibrium 2.00 6.00 4.00 Complete sample problems 6 & 7 for more practice using ICE charts.
Equilibrium Constant in Terms of Pressure • Equilibria involving gases can be described in terms of either pressure or concentrations. • The relationship can be seen using the Ideal Gas Law, PV=nRT. • Rearranging this equation gives • n/V represents concentration in M. Complete sample problem # 8 now.
K vs. Kp • For any reaction: Kp = K(RT)Dn • Dn = sum of coefficients of gaseous products minus sum of coefficients of gaseous reactants. Complete Sample Problem #9.
Reaction Quotient • After the equilibrium constant (K) is known, we can use it to determine if a reaction is at equilibrium. • The reaction quotient, Q, has the same form as the equilibrium constant expression EXCEPT initial concentrations are used instead of equilibrium concentrations. • H2(g) + F2(g) « 2HF(g)
Predicting the Direction of a Reaction Using Reaction Quotient • If Q > K then the reverse reaction must occur to reach equilibrium (i.e., products are consumed, reactants are formed, the numerator in the equilibrium constant expression decreases and Q decreases until it equals K). • If Q < K then the forward reaction must occur to reach equilibrium. • If Q = K then the reaction is at equilibrium.
Practice Problem Part 2: • Using the previous reaction: • Fe3+(aq) + SCN-(aq) FeSCN2+(aq) and the K value we determined: K = 0.33 determine if the following concentrations are at equilibrium: • Initial:10.0 M Fe3+(aq), 8.00 M SCN-(aq), and 2.00 M FeSCN2- Complete sample problems 10 & 11.
Solving Equilibrium Problems • 1. Balance the equation. • 2. Write the equilibrium expression. • 3. List the initial concentrations. • 4. Calculate Q and determine the shift to equilibrium.
Solving Equilibrium Problems(continued) • 5. Define the change needed to reach equilibrium. • 6. Substitute equilibrium concentrations into equilibrium expression and solve. • 7. Check calculated concentrations by calculating K. Complete more complex sample problems #12-15.
Le Châtelier’s Principle • . . . if a change is imposed on a system at equilibrium, the position of the equilibrium will shift in a direction that tends to reduce that change.
Le Châtelier’s Principle • Consider the production of ammonia • As the pressure increases, the amount of ammonia present at equilibrium increases. • As the temperature decreases, the amount of ammonia at equilibrium increases. • Can this be predicted? • Le Châtelier’s Principle: if a system at equilibrium is disturbed, the system will move in such a way as to counteract the disturbance.
Effects of Changes on the System 1. Concentration: The system will shift away from the added component. 2. Temperature: K will change depending upon the temperature (treat the energy change as a reactant).
Effects of Changes on the System (continued) 3. Pressure: a. Addition of inert gas does not affect the equilibrium position. b. Decreasing the volume shifts the equilibrium toward the side with fewer moles.
Le Châtelier’s Principle • Change in Reactant or Product Concentrations • Consider the Haber process • If H2 is added while the system is at equilibrium, the system must respond to counteract the added H2 (by Le Châtelier). • That is, the system must consume the H2 and produce products until a new equilibrium is established. • Therefore, [H2] and [N2] will decrease and [NH3] increases.
Le Châtelier’s Principle Change in Reactant or Product Concentrations
Le Châtelier’s Principle The Haber Process
Le Châtelier’s Principle The Haber Process for producing NH3 • N2 and H2 are pumped into a chamber. • The pre-heated gases are passed through a heating coil to the catalyst bed. • The catalyst bed is kept at 460 - 550 C under high pressure. • The product gas stream (containing N2, H2 and NH3) is passed over a cooler to a refrigeration unit. • In the refrigeration unit, ammonia liquefies but not N2 or H2. • The unreacted nitrogen and hydrogen are recycled with the new N2 and H2 feed gas. • The equilibrium amount of ammonia is optimized