Hematopoiesis: Basic Concepts
Hematopoiesis: Basic Concepts Blood cell production is highly regulated to maintain circulating cell numbers within relatively constant levels and to respond rapidly to conditions requiring extra cells

Hematopoiesis: Basic Concepts
E N D
Presentation Transcript
Hematopoiesis: Basic Concepts • Blood cell production is highly regulated to maintain circulating cell numbers within relatively constant levels and to respond rapidly to conditions requiring extra cells • Continuous and prodigious production in the adult marrow and lymphoid tissues of new, mature blood cells from more primitive precursors: ~1011 per day (< 1010 RBCs/hour, 108- 109 WBCs/hour) • Normal hematopoiesis involves an exquisitely regulated balance between self-renewal, terminal differentiation, migration, and cell death. • Replacement of hematopoietic cells is achieved via differentiation of primitive pluripotent stem cells through a series of cell divisions (usually the last 3 to 5 cell cycles exhibit terminal differentiation programs of most lineages)
Hematopoiesis: Basic Concepts • All arise from a single cell type – the hematopoietic stem cell (HSC). • HSCs are pluripotent (can give rise to differentiated blood cells of all lineages – T & B & NK lymphocytes, erythrocytes, mast cells, megakaryocytes & platelets, eosinophils, basophils, neutrophils, monocytes/macrophages, DCs, etc.). Lack the markers of differentiated blood • HSCs are rare, mainly quiescent, undifferentiated cells that on occasion produce by mitosis 2 kinds of progeny: • more stem cells (self-renewal) • cells that begin to differentiate along the various paths to terminally differentiated hematopoietic lineages • Path of differentiation is generally regulated by the need for more of that particular type of blood cell, and is controlled by the appropriate cytokines and hormones and growth factors, colony stimulating factors (CSFs)
Model of Stem Cell Decisions: HSC will choose one of 2 pathways: self-renewal (maintains primitive state) or differentiation (driven toward a more mature state)
lymphocytes – terminally diff.: can undergofurther division (i.e. memory cells) Granulocytes – terminally diff.: no further division anuclear
Lineage diagram – outline of how hematopoietic cells are increasingly restricted in the types of progeny to which they can give rise as differentiation proceeds
The Hematopoietic Developmental Hierarchy • During the life of an individual, 2 separate hematopoietic systems • Both arise during embryonic development but only one persists in adult • PRIMITIVE HEMATOPOIESIS– system derived from the extra- • embryonic YOLK SAC; consists mainly of nucleated erythroid cells • that carry oxygen to the developing embryonic tissues… an early • circulatory system. • As the embryo’s size increases, primitive system superceded by • DEFINITIVE HEMATOPOIETIC system, which originates in the • embryo itself and continues throughout adult life. Is made up of all • adult blood cell types including erythrocytes, and cells of myeloid • and lymphoid lineages.
Sites of hematopoiesis: During development, hemato- poiesis is initiated sequentially in different tissues PRIMITIVE HEMATOPOIESIS occurs in the YOLK SAC at mouse embryonic day 7.5 (E7.5), and probably starts ~ 4 weeks in humans Primitive hematopoiesis is characterized by the productionof fetal erythrocytes (nucleated) and the lack of lymphocytesand myeloid cells except for macrophages. For many years the YS was assumed to be the primary siteof formation of the HSCs that migrateto and colonize the fetal liver and subsequently the bone marrow. d. 31-34
1stDEFINITIVE multipotent hematopoietic stem cells are generated within the embryonic AGM region of the para-aortic splanchnopleuric mesoderm (d. 30-37 human, and late d. 10/early d. 11 mice) DEFINITIVE HEMATOPOIESIS – HSCs can restore long-term multilineage hematopoiesis when transplanted into adult myeloablated recipients, and generates enucleated erythrocytes, various kinds of myeloid andlymphoid cells, and long-term reconstituting hematopoietic stemcells (LTR-HSCs)… Upon transplantationinto NOD-SCID mice, cultured AGM HSC cells showed lymphomyeloidreconstitution; YS cells were only capable of contributingto the myeloid lineage (Tavian et al., 2001). CD34+ cluster of cells (also CD45+ hemato-specific) AGM region – aorta-gonad-mesonephros ventral floor of dorsal aorta (also umbilical and vitilline arteries @ connection with dorsal aorta)
Source of these first definitive HSCs?? Shared expression patterns of a number of molecules by both intra-aortic cluster HSCs and underlying endothelial cells supports the existence of a HEMANGIOBLAST or primitive mesenchymal endothelial-like cell with hematogenic potential that lies on the ventral floor of the dorsal aorta… AKA “hemogenic endothelium” Example: VEGF receptor (VEGF R): neither HSCs nor blood vessel epithelium develop without the ligand, VEGF. Also, angiopoietin: produced by early blood cells to induce blood vessels to grow in the vicinity. Recent ID of a morphologically distinct layer of cells resembling a stromal layer underlying the ventral floor of the dorsal aorta in the AGM suggests that this region could represent a microenvironment or niche supporting HSC development (Marshall et al., 1999, Dev. Dyn. 215:139)
The fetal liver is the main organ for hematopoiesis during fetal life After about d. 37 (5 weeks), HSCs from the AGM begin to colonize the fetal liver, and at 6 weeks hematopoiesis (definitive) takes place inthe fetal liver until bone marrow isformed Throughout fetal life, the liver is the chief organ for production of myeloid and erythroid cells.
Fetal liver HSCs seed thymus, bone marrow and spleen At about 8 wks, liver HSCs differentiate in the thymus to mature T lymphocytes (~10 wks) which populate fetal lymph nodes, spleen and gut by 12 wks and other peripheral lymphoid tissue by weeks 14-15. Bone marrow (BM) is seeded by liver HSCs by 8 weeks. B-lymphopoiesis takes place in liver @ 7 wks., then shifts to bone marrow. Hematopoiesis from fetal BM is mainly myeloid and contributes only minimally to the blood pool throughout fetal life. After birth, BM becomes main hematopoietic organ. Many HPC and HSC in circulation during fetal life and immediately after birth; 24-48 hours after birth they disappear due to lodgement in BM.
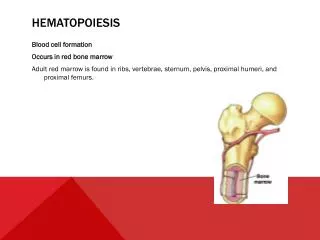
Bone marrow: site of pre- and peri-natal, childhood, and adult hematopoiesis During fetal growth, hematopoiesis takes place in all bony cavities (axial and appendicular skeleton) as well as in liver and spleen. Prior to birth, splenic and hepatic hematopoiesis disappear, and gradually thereafter hematopoietic tissue (red marrow) is replaced by adipocytes (yellow marrow) beginning in the distal bones and retracting to the adult pattern by age ten. Yellow marrow can be reactivated by an increased demand for blood cells, i.e., blood loss, but does not normally produce blood cells In the adult, hematopoietic marrow is confined to the axial skeleton (sternum, vertebrae, iliac bones, ribs) and proximal portions of the humerus and femur.
Bone marrow, cont’d Bone marrow is specially construed to support the proliferation, differentiation, and maintenance of hematopoietic cells Honeycombed latticework of venous sinuses – large, thin-walled veins Endothelial cells lining the marrow sinuses are bounded by STROMAL CELLS that generate an EXTRACELLULAR MATRIX that mechanically support hematopoietic cells and vasculature; provide a nurturing microenvironment for hematopoiesis & hematopoietic colonies Adjacent cells in marrow: endothelial cells, fibroblasts, adipocytes, osteoblasts, and macrophages and reticular connective tissue. Close contact between hematopoietic cells and these cells, especially the stroma facilitates transmission of proliferative signals or diffusion of locally produced cytokines Maturing blood cells can enter the circulation through openings in the vascular sinuses (megakaryocytes & erythroblasts clustered against sinuses) – usually go first to other hematopoietic tissues for further maturation Distinct microenvironments per lineage, i.e., erythropoietic, eosinophilic, etc.
Bone marrow, cont’d • BONE MARROW STROMAL CELLS: Stroma – derived from Greek word meaning mattress or bed. • Large spread-out cells that appear to provide a bed for hematopoietic cells • Derived from mesenchymal stem cells and are not of hematopoietic origin. • Express class I histocompatibility antigens, but lack the hematopoietic cell surface marker CD45 • Attempts to classify stromal cells by morphology have resulted in a multitude of descriptive names, including adipocytes, pre-adipocytes, smooth-muscle-like, fibroblastoid, endotheloid, epitheloid, blanket, and reticulum cells. • So far, there are no definitive markers that predict whether a stromal cell line will support stem cells. Neither the morphology of stromal cells, nor the known cell surface antigens, nor the patterns of cytokines production are predictive of support function.
From NIH Stem Cell Primer at http://www.nih.gov/news/stemcell/scireport.htm
The hematopoietic stem cell • All hematopoietic cells arise from a single type of cell – the hematopoietic stem cell (HSC). • HSCs are pluripotent (can give rise to differentiated blood cells of all lineages – T & B & NK lymphocytes, erythrocytes, mast cells, megakaryocytes & platelets, eosinophils, basophils, neutrophils, monocytes/macrophages, DCs, etc.) • HSCs are rare (1 in every 105 nucleated cells in adult bone marrow) • Are mainly quiescent, undifferentiated cells that on occasion produce by mitosis 2 kinds of progeny (asymmetric division) : • more stem cells (HSCs have a limited ability to self-renew) • progenitor cells that can undergo further divisions and become progressively more differentiated and more restricted in their capacity for self renewal
The hematopoietic stem cell, cont’d • Express the CD34 surface marker, but lack differentiation markers of more mature cells (such as CD38). Often express the receptor for vascular endothelial growth factor (VEGF). Also express Sca-1. • Because they are mitotically quiescent, can also be selected on their ability to survive in presence of 5 fluorouracil (5FU) which kills proliferating cells. • In vivo, HSCs (as well as the full hierarchy of hematopoietic progenitor cells) are maintained throughout adult life… constant, very slow state of turnover. • In culture, in the absence of the appropriate regulatory signals, tend to lose ability to self-renew and become lineage-restricted • In vivo, a highly regulated microenvironment ensures that pluripotent stem cells are generated in sufficient numbers and at the appropriate developmental stage to seed subsequent hematopoietic tissues
Functional assay to ID stem cells in vivo: is the cell able to stably generate multiple hematopoietic lineages in irradiated or SCID (immunodeficient) mice? • Have done this with human HSCs, termed “SCID repopulating cells” or SRCs… able to repopulate human hematopoiesis in immunodeficient mice -- LTRlong-term repopulation • SRC homes to & engrafts murine BM and produces immature cells • Capable of multilineage diff’tn • Only found in CD34+/38- frxn • -- purify these to get enriched • HSC population, and/or select • for VEGF receptor expression • After cells are transplanted, a • CD34+/CD38- population of • cells supports short term • hematopoiesis, following which • a CD34-/CD38- population • supports long-term reconstitution
XY XX Sacrifice, take marrow 48 hrs XX XY Short-term reconstituting XX blood cells XX months Blood is XY Most strict definition of an HSC: ability to serially reconstitute hematopoiesis (HSC loses ability to do this after it has self-renewed several times) HSC home to marrow & are quiescent for up to 48 hrs Serial transplantation to 2ndary host
Progenitor cells and precursor cells • Cycling stem cells renew and give rise to more mature multipotent progenitor cells, which are more restricted in the offspring which they will generate. This is associated with tremendous amplification in cell number. • Progenitor cells • are multipotent • do not self-renew or have only an extremely limited capacity • respond best to multiple cytokines • is a compartment of hematopoiesis that expands the number of cells dramatically • are named by the types of colonies they give rise to: • The pluripotent HSC gives rise to lymphoid and myeloid stem cells, the latter of which gives rise to a GEMM progenitor termed CFU-GEMM • CFU-GEMM is a multipotent cell giving rise to granulocyte, erythroid, monocyte, and megakaryocyte colonies • CFU-GM gives rise to both granulocyte and monocyte colonies
Self renewal Proliferation X Differentiation Expression of new sets of genes • Cell cycle status is tightly associated with the function of cells at each step of hematopoiesis: • Primitive stem cells (CD34+) – slow cell cycling or dormancy • Progenitor populations – rapid cycling required for effective expansion • Terminally differentiated cells – withdrawal from cell cycle suitable and sometimes prerequisite for functions of mature cells
Progenitor cells and precursor cells • Precursor cells (“Committed” precursor cells) • blast cells committed to unilinear differentiation – much more mature than progenitor cells • do not self-renew • respond best to one or 2 cytokines • still replicate until near terminal differentiation • progeny increasingly acquire specific differentiation markers and functions • include CFU-G, CFU-M, CFU-E, and CFU-Baso, giving rise respectively to granulocytes, monocytes, eosinophils, and basophils
Self renewal Proliferation X Expression of new sets of genes Differentiation • Cell cycle status is tightly associated with the function of cells at each step of hematopoiesis: • Primitive stem cells (CD34+) – slow cell cycling or dormancy • Progenitor populations – rapid cycling required for effective expansion • Terminally differentiated cells – withdrawal from cell cycle suitable and sometimes prerequisite for functions of mature cells
Differentiation and lineage commitment • Path of differentiation is generally regulated by the need for more of one particular type of blood cell, and is controlled by the appropriate cytokines and hormones and growth factors, colony stimulating factors (CSFs) • Differentiation and lineage commitment occur under the influence of a complex array of signals from the extracellular environment, especially cytokines such as stem cell factor (c-Kit ligand), IL-3, GM-CSF, and G-CSF – binding of cytokines to cell surface receptors results in the initiation of a cascade of signal transduction events within the cell. • Series of negative regulators & amplification circuits provide additional control over the process of hematopoiesis.
Neutrophils •Major component of innate immune system… 1st line of defense against infection •Surround microorganisms with pseudopodia •Pseudopod fusion to form phagosome (phagocytosis) •Granuole release into phagosome •Secretion of granuole contents
Francois Paraf, M.D NEJM 1997
A key concept : The marrow contains a large storage pool of neutrophils which can be reserved for release in a setting of stress, and that the exponential expansion of progenitor cells can be augmented by granulocyte colony stimulating factor (G-CSF) under stress conditions. • Neutrophils and Host Defense • • Most are in the marrow--reserve • • Circulating neutrophils: half in vessel, half adherent to wall • • Have peptide receptors for and diapedese in response to FMLP (N- formyl-methionyl-leucyl-phenylalanine),chemotaxins • Steady flow of neutrophils into superficial tissue, skin, mucosa, lungs needed to prevent infection. • • Bactericidal mechanisms • – Degradative enzymes in granules • – Oxidative killing • • Superoxide generation by NADPH-dependent oxidase • Involves hexose monophosphate shunt, cytochrome b • Patients with Chronic Granulomatous Disease lack cytochrome b cannot generate superoxide, and develop repeated infections.
9 X 109 cells/kg in post-mitotic pool 2.6 X 109 cells/kg in mitotic pool Example of differentiation from the precursor stage onward: myeloblast to granulocyte (neutrophil maturation) Terminal cell division Increasing development of granuoles (antibacterial and phagocytic) Increasing phagocytic function – pseudopodia extend around microorganisms and fuse to form a phagosome into which granuole contents are released Expansion of cell number occurs as cells in the mitotic or proliferative pool replicate The post-mitotic pool can no longer divide but continues to mature into terminally differentiated cells In the setting of infection or stress, maturation time may be shortened, divisions may be skipped, and cells may be released into the bloodstream earlier
Mobilization of marrow cells (transendothelial migration) Mobilization of hematopoietic progenitor cells is a multistep process – common themes in this process (not in chronological order): 1) Adhesion interactions -- must first be disrupted for progenitor cell trafficking. In general adhesion molecules are expressed on hematopoietic progenitor cells in the marrow but are downregulated or degraded to facilitate egress of progenitors from the BM -- selectins, selectin ligands, integrins, CD44 and PECAM 2) Chemotaxis Transendothelial gradients of chemokines (cytokines with chemotactic activity) produced by stromal cells control direction and efficacy of transendothelial migration of hematopoietic progenitors – mature leukocytes respond to a variety of chemokines, but most important so far is SDF-1 stromal cell-derived factor-1 (made by BM stromal cells). Significant chemotactic activity in hematopoietic progenitor and stem cells…significantly enhances retention in BM and homing to BM. The chemokine receptor CXCR-4 is the receptor for the SDF-1 chemokine similarly upregulated before mobilization and downregulated after transendothelial migration. 3) Paracrine cytokines may support mobilization – Growth factor stimulated hematopoietic cells produce cytokines such as vascular endothelial growth factor (VEGF) that act on endothelial cells to support migration by increasing endothelial fenestration and permeability.
Mobilization of stem cells for HSCT Mobilization – various molecules administered to donors can mobilize CD34+ stem cells out of marrow into circulation where can harvest from peripheral blood… granulocyte-colony stimulating factor (G-CSF), granuloctye-macrophage colony stimulating factor (GM-CSF), flt-3 ligand, stem cell factor (SCF), and a variety of cytokines (IL-7, IL-3, IL-12) and chemokines (IL-8, SDF-1), as well as chemotherapeutic agents cyclophosphamide and paclitaxel, with varying degrees of efficacy G-CSF (Neupogen®, Filgrastim®) most commonly used, daily stimulations of healthy donors, sometimes used with the chemotherapeutic agent cyclophosphamide – induce proliferation of hematopoietic cells within the bone marrow Mobilized PBL cells have a much faster engraftment than do bone marrow cells, due to the increased cell dose of transplanted mobilized cells and increased numbers of committed progenitor cells. BUT – proliferation & differentiation potential of CD34+/CD38- stem cells from mobilized PBL is inferior to that of undifferentiated BM. Cord blood stem cells: Immediately after birth, relatively high levels of immature CD34 + progenitor & stem cells circulating (for about 48 hours)… umbilical cord blood represents a promising source of stem cells for transplantation, but #s are too low for transplant into adults – need to find a way to expand ex vivo (stromal cells).
Model of mechanisms of stem cell mobilization by G-CSF: disruption of retention in BM & proximity to stromal cells • 1) Disruption of adhesion interactions btwn. HSC and BM stromal microenvironment • VLA-4 is an integrin on HSCs that binds to VCAM-1 (another adhesion molecule) on stromal cells -- needs to be downregulated for HSCs to egress. HSCs also increase the expression of proteolytic enzymes elastase MMPs, and cathepsin G during mobilization so that they can cleave VCAM-1 from the stromal cells. • Disruption of chemotactic interactions • SDF-1 retention signal produced by BM stromal cells gets degraded by same proteolytic enzymes • CXCR4 is upregulated on HSCs in mobilization… why? Probably important for egress to interact with SDF-1 in the blood. • 3) Degradation of extracellular matrix Lapidot and Petit, 2002 Exp. Hematology 30:973