Epigenetics
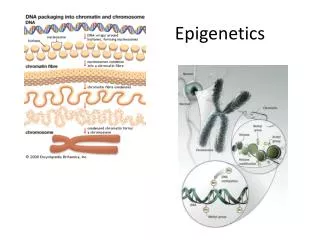
Epigenetics. A nucleosome is the basic unit of DNA packaging in eukaryotes, consisting of a segment of DNA wound in sequence around eight histone protein cores. This structure is often compared to thread wrapped around a spool.

Epigenetics
E N D
Presentation Transcript
A nucleosome is the basic unit of DNA packaging in eukaryotes, consisting of a segment of DNA wound in sequence around eight histone protein cores. This structure is often compared to thread wrapped around a spool. • Nucleosomes form the fundamental repeating units of eukaryotic chromatin, which is used to pack the large eukaryotic genomes into the nucleus while still ensuring appropriate access to it (in mammalian cells approximately 2 m of linear DNA have to be packed into a nucleus of roughly 10 µm diameter).
A nucleosome core has about 146 bp of DNA • Typical exon of around 140 nt
Epigenetics • Epi- (Greek: over, above, outer) • The study of mitotically (separating chromasomes in a cell) and/or meiotically heritable changes in gene function that cannot be explained by changes in DNA sequence (96 Russo)(classical) • 11_Graff(www.cs.uml.edu/~kim/580/11_Graff.pdf) • Structural adaptation of chromosomal regions so as to register, signal, or perpetuate altered activity states (recent) • Study of changes in gene expression or cellular phenotype (simple)
Three Major Levels of Epigenetic Changes • Chemical modifications at the level of nucleotides • Including DNA methylation and RNA interference • Modifications at the level of histones that encompass posttranslational modifications (PTMs) of histone proteins and the incorporation of histone variants • Nucleosome remodeling • ATP-dependent processes that regulate the accessibility of nucleosomal DNA • => Regulation of the accessibility of the chromatin structure to the transcription machinery
1. a. DNA Modification: Methylation • Covalent addition of a methyl group from methyl donor SAM (S-adenosylmethionine) to a cytosine base • Occurs mainly at 5’ end of cytosine in CpG, CpHpG and CpHpH, where H is A,T, or C • This reaction is catalyzed by a family of DNMT (DNA methyltransferase) • DNMT1 is the main enzyme in mammals • Methylation patterns change over evolution • In invertebrate animals, mosaic methylation, with stable methylated domains interspersed with methylation-free regions • In vertebrate genomes, globally methylated, with exception of CpG islands • Methylation dynamically change among different cells, and even in a single cell
The reaction catalyzed by DNA methyltransferases (DNMTs). DNMTs are the key enzymes for DNA methylation and catalyze the transfer of a methyl group from SAM to cytosine, thus forming 5-methyl-cytosine and SAH. Methylation of CpG sequences might induce chromatin conformational modifications and inhibit the access of the transcriptional machinery to gene promoter regions, thus altering gene expression levels. Therefore, promoter rmethylation of CpG islands is commonly associated with gene silencing and promoter demethylation with gene expression, though several exceptions to this rule are known.
MethylationInheritance • Both C & its complementary G are methylated (fully methylated) • After replication, rapidly acted on by DNMT1 to regenerate two identical fully methylated double helices • Epigenetic info is inherited in the form of DNA methylation patterns
DNA Methylation Binding • Methyl-binding proteins (MBPs) bind to methylated DNA, typically in promoters (e.g. MeCP2– Methyl CpG binding Protein 2) • Binding recruits other protein complexes that lead to transcription repression
1.b. RNAi • Epigenetic alterations of DNA can also be produced by double-stranded RNA (dsRNA) and protein components of RNAi machinery • Small RNAs produced by cleavage of dsRNA are thought to serve as sequence-specific facilitators to guide other enzymes of epigenetic machinery into place • D.melanogaster • Members of RNAi machinery such as genes piwi and homeless are mutated, centromeric heterochromatin formation is inhibited • Fission yeast (Schizosaccharomyces pombe) • Deletions of genes involved in RNAi machinery, such as argonaute, result in reduced hetrochromatin formation and reduced methylation on H3K9 (marker of gene repression) • Mammals • Short-interfering RNA (siRNA) induce methylation alongside H3 methylation, resulting in decreased gene expression
2. a. Histone Modification • Histone • Basic proteins regulating compaction of chromatin • Consist of • a loosely structure NH2-terminal tail • out acting as regulatory substrates for nucleosomal stability • These substrates establish condensed/uncondensed states of the chromatin • a globular histone core (nucleosome) • Octamer of four core histones H2A, H2B, H3, and H4 in duplicates • Around the core, 147 bp wrap around in 10-nm-thick primary structure
2. a. Histone Modification • Histone • Nucleosomes are linked together by linker histone H1 • Post Translational Modifications (PTMs) can occur on all histones • Majority occurs on NH2-terminal tail • PTM types • Acetylation • Methylation • Phosporylation • Ubiquitnation • sumoylation
DNA-gray H2A – blue H2B – yellow H3 – green H4 - red
PTM • Their accessibility of DNA will change • Current research emphasis is on • Role of modification in the transformation process from normal to cancer cells • Study imbalances in net expression of tumor suppressor vs. oncogenes or overall genomic imbalances • Needs substrate specificity and residue-specific alteration
Acetylation • Positively charged AA (lysine(K) and arginine (R) are neutralized by acetyl group, leading to decreased affinity between histone tail and negatively charged DNA • HAT (Histone Acetyltransferase) regulates acetylation of histones • In most cancers, HAT genes are muted and HAT includes chromosomal translocation of respective HAT • But HDACs (Histone deacetylase) are frequently overexpressed • Histones prefer being methylated or phosphorylated at R, at acetylated at K • Acetylation of K initiates active gene expression • Acetylation plays a role in nucleosome assembly and maintenance of chromatin state affecting DNA repair, etc.
The unmodified side chain of posttranslationally modifiable residues is first presented, followed by representations of the residue on which the posttranslational modification has occurred at respective sites. Abbreviations are as follows: K, lysine; R, arginine; Y, tyrosine; S, serine; T, threonine; -ac, acetylation; -me, monomethylation; -me2, dimethylation; -me3, trimethylation; -ph, phosphorylation. The color code is as follows: yellow, carbon; blue, nitrogen; pink, polar hydrogen; red, oxygen; orange, phosphorus; green, methyl groups of posttranslational modifications.
Effects of acetylation and methylation on histone residues. The red circle represents acetyl groups, the yellow circle symbolizes methylation and the green symbolizes methylation of arginine residue. Acetylation of lysine residues is associated with gene expression whereas methylation-mediated expression is dependent on the residue methylated and the position. These reversible processes exerted by epigenetic enzymes HATs, HDACs, and HMTs largely affect genomic stabilities, local gene expression, and factors governing cell fate or phenotype. This figure is reproduced in the color plate section.
Effects of acetylation on protein functions. Acetylation of proteins affects many different functions, some of which are listed. The double up-arrows indicate increase and the double down-arrows indicate decrease with respect to the particular function. Some of the genes affected by acetylation under specific protein functions are listed [60].
Methylation • K and R can be mono-, di- or tri-methylated forms • Monomethylated H3K4 is found in expressed and repressed genes • Trimethylated H3K4 is exclusively in silenced genes • Location – H3K9me in coding region for expression, in promoter, repression • Some times, same K is acetylated or methylated • example: K4 and K9 residues of histone H3 • Methylation is regulated by • HMT (histone methylatransferase), specific to K and R • And HDM (Histone Demethylase)
Methylation 2 • Common pattern in many cancers • Loss of H4K16 acetylation and H4K20 tri-methylation • When tumor suppressor genes are down-regulated by hypermethylation, oncogenes may be stimulated by acetylation or hypomethylation • Example: hypermethylation of H3K79 promotes leukemogenesis • Tumor-specific epigenetic abnormalities can stem from altered modifications of the histone residues, and/or altered expression of the enzymes that catalyze the modifications
Methylation 3 • Histone lysine resideus are methylated by methyltransferases and utilizes S-adnosyl methionine (SAM) in catalyzing the transfer of methyl group to specific histone residues • Methyltransferases are specific based on target residues • PKMT (Protein lysine methyltransferase) for lysine • PRMT (Protein Arginine MT) for R • PRMT primarily catalyze mono- and di-methylation of histone R 2,8,17, 26 of H3, and R 3 of H4 • H3K27 methylation is mediated by a PKMT called EZH2, which is over-expressed in many tumors and considered to be responsible for cancer aggressiveness • Leukemogenesis is promoted by aberrant recruitment of H3K79
Role of Methylation • Gene silencing (exceptions are found) • Maintaining cellular functions and development of autoimmunity and aging • Aberrant methylation • may be associated with disorder of gene expression • Irregular memory function in development by heritability • Reversible process • Demethylation by enzymes such as DNA glycolaes
2.b. Histone Variants • Results from sequential and structural variation of core histones • Replacement of large groups of AAs in histone tails and globular central domains • Only a few AA substitutions • Four core Histones are incorporated into nucleosomal structure exclusively during replication • Histone variants can be integrated into specific regions of genome throughout cell cycle
3. Nucleosomal Remodeling • Chromatin structure is changed from net energy input • Nucleosome remodeling is carried out by enzymes that are catalytically dependent on ATP as energy source
Gene regulation • 12_Su • Chromatin components mainly include • Histone modifications • Histone variants • DNA-binding proteins and associate complexes • In mammalian genomes, chromatin components and DNA methylation are associated with chromatin regulation, and influence gene transcription • How they regulate chromatin structure and gene expression has implications for understanding development, aging and disease • Most histone modifications occur at the flexible N-terminal tails
Gene regulation 2 • Histone acetylation – gene activation • Histone methylation – gene activation and repression • example. • Enrichment of H3K9ac and H3K4me3 in promoters and CpG islands are associated with gene repression • Histone variant H2A.Z and RNA pol-II are preferentially deposited in promoters, yielding gene activation
Gene Expression • How chromatin components and DNA methylation affect gene expression? Independently or synergistically ? • Classify genes into HGP (High-expression gene promoters) and LGP • Modification intensities except H3K9me2 and H4K20me3 are significantly distinct between HGPs and LGPs
Chromatic Structure Alteration • Alteration of chromatin fiber structure is critical to control cellular processes and regulate the expression fidelity of genes in particular cell types • Such regulation is carried out by a combination of several factors, posttranslational histone tail modification, chromatin remodeling enzymes. • One factor contributing this process comes from architectural proteins such as H1 and members of HMG (high-mobility-group) superfamily • HMG superfamily – HMGA, HMGB, HMGN • HMGN – unique in its ability to bind directly to the nucleosome core particles • HMGN – associated with generation and maintenance of open chromatin regions
HMGN1 is enriched in transcriptionally active domains. Transcriptionally inactive chromatin is marked by H3K27me3. Active chromatin marks such as H3K4me3 and H3K9ac, is seen close to the gene Actrively transcribed with RNA Pol II across promoter
Alzheimer’s Disease • AD • Neurodegeneration in brain regions including temporal and paretal lobes and restricted regions in frontal oortes and cingulate gyrus • Exracelluar amyloid deposits (senile plques, SP) and the presence of neurofibrilliary tangels (NFT) composed of intraneuronal aggregates of hyperphosphorylated tau protein • Primary component of SP is about 40 bp amyloid β (Aβ), resulting from proteolytic processing of its precursor, amyloid precursor protein (APP) • APP is processed by β- and γ-secretase (presenilin and other protein complex) to produce Aβ: Aβ40, Aβ42 • A high Aβ42/Aβ40 => AD
AD • 1% early AD • 50% due to mutations in APP, PSEN1, PSEN2 • 50% may involve other • LOAD (Late-onset AD) over age 65 • ALZGene database (alzgene.org) lists over 1000 genes • Most like genes • APOE (apolipoprotein E) • BIN1 (bridging integrator 1) • CLU (clusterin) • Found to have decreased folate values and increased plasma homocysteine levels (hyperhomocysteinemia)
One-carbon metabolism • Folate • Essential nutrients required for one-carbon biosynthetic and epigenetic process • Derived entirely from dietary sources, mainly from green vegetables, fruits, cereals, and meat • After intestinal absorption, folate metabolism requires reduction and methylation into the liver to from 5-methylterahydrofolate (5-MTHF), release into blood and cellular uptake • 5-MTHF is used for synthesis of DNA and RNA precursors or for conversion of homcyctein (Hcy) to methionine, which is used to form S-adenoylmethionine (SAM) • B6 and B12 participate in one-carbon metabolism • Folic acid is the syntheic form
One-carbon (Folate) Metabolism • High level of Hcy is suspected
One-carbon (Folate) Metabolism • DNA methylation is dependent on its potential measured by SAM/SAH level • High SAH level inhibit DNMTs