Understanding Speed, Forces, and Energy in Physical Science
290 likes | 426 Views
This comprehensive outline covers the fundamental principles of speed, force, acceleration, and energy in physical science. It explains key concepts such as constant speed, acceleration, Newton's laws of motion, work, and energy transformations, including kinetic and potential energy. Each section provides various examples and answers to reinforce understanding. Ideal for students looking to deepen their knowledge of physics concepts and prepare for assessments.

Understanding Speed, Forces, and Energy in Physical Science
E N D
Presentation Transcript
EOC Physical Science Goal 2
Goal 2 • 1. Speed = Distance/Time • 3/1=3 6/2=3 9/3=3 12/4=3 • Answer =C • Constant speed graph • 2a. Constant speed forward • 2b. Acceleration • 2c. Constant speed backward • 2d. Deceleration • Answer=B
Goal 21 • 3. Answer = D • 4. v=d/t v= 12m/s, t=3s 12 x 3=36m Answer = D • 5. a=vf-vi/t vf=40 vi=0 t=5 40-0/5=8m/s/s Answer B • 6. 0-3s=constant speed, 3-10s slower constant speed, 10-15s stopped, 15-17s=fastest constant speed From 0-5s 6m was covered so 6/5= 1.3m/s Answer = B • 7. A= V/t 34/1.7=20s Answer =C • 8. A=constant speed, B=stop, C=Acceleration, D=Stop Answer=C V A T
Goal 2 • 9. Change in force causes a CHANGE in speed or direction. The load is moving at a constant speed the forces are balanced not unbalanced. Answer=C • 10. Same acceleration of gravity at 9.8m/s • Answer = C • 11. F=ma 3 x 2 = 6N Answer =D • 12. M=500kg , A=4m/s/s 500x4=2000N Answer=C • 13. Equal force opposite direction=balanced forces so object will not move Answer=A F M A A
Goal 1 • 14. The force is on the card not the coin. Because of inertia the coin will drop into the cup. Answer=C • 15. F=MA Cart W 3/5=0.6, • Cart X 6/5=1.2 • Cart Y 4/5=0.8 • Cart Z 1/5=0.2 • Answer=B • 16. f=100N, A=2m/s/s 100/2=50 Answer B F M A
Goal 2 • 17. Equal and opposite reaction pull cart toward he moves back Answer C • 18. Balanced forces because chair is not accelerating so the answer is B • 19. Newton’s 3rd law, action/reaction Answer is C • 20. Gravity is pushing down on the table and the floor is pushing back with an equal force so the answer is A.
Goal 3 • 1. Kinetic Energy=motion Where is the cart moving the fastest Answer = B The point with the greatest potential energy would be W because it is the highest point. • 2. ½(m)(v)² ½(1)(5)²=12.5 J Answer is C • 3. mgh m=5, g=9.8, h=5 5(9.8)(5)=245J Answer=C • 4. Density is mass per unit volume • Specific Heat is the amount of energy to move temperature 1ºC • Melting point temperature substance goes from solid to liquid • Boiling point temperature substance goes from liquid to gas • Answer=B
Goal 3 • 5. Ethyl alcohol has a lower specific heat than water so it will change temperature quicker than water Answer=A • 6. PE=mgh m=3 g=9.8 h=10 (3)(9.8)(10)=294J Answer=C • 7. P=w/t P=fxd/t f=50, d=10, t=10 50x10/10=50W Answer=B • 8. W=750J F=125N 750/125=6m Answer =A W F D
Goal 3 • 9. W=FxD P=W/t W=500 x 10=5000J P=5000/4=1250W Answer=D • 10. For work to occur the force has to be in the same direction as the work and a distance must be covered. No work is done in A because no distance is being covered. • 11. Heat moves from High to low so from the water to the solid so the solid temp will increase the water temp will decrease until they are both the same temp.
Goal 3 • 12. Heat will move from the water to the aluminum until they reach equilibrium. Answer = C • 13. Heat flows from the hot drink to the ice. Answer D • 14. Light travels faster than sound. Answer B • 15. The air is cycling, heat rising, cool falling Answer= D Conduction=direct contact, radiation=waves through space • 16. Answer C Look at EMS • 17. Energy=Amplitude or Height Answer-D
Goal 3 • 18. frequency= short wavelength, low energy= low amplitude Answer=A • 19. Volume=Amplitude=Energy Pitch= how high or low tone is AnswerD • 20. V=λf λ=0.5, f=20 0.5 x 20 = 10m/s
Goal 4 • 1. Electrons flow from high concentration to low. The rod becomes negatively charged so it pulls electrons off of the fur. Answer=D • 2. Electrons flow from the place where there are more of them to the place with less when objects touch. The answer is A • 3. Opposites attract, like charges repel. Answer is A • 4. The positive charges on the paper move toward the balloon attracting them to the balloon. Because this happened without direct contact it is induction. Answer is A
Goal 4 • 5. V=IR V=6 volts, R=3Ω 6/3=2A Answer=B • 6. P=IV P=45W I=0.5A 45/0.5=90V Answer B • 7. P=650W V=120V 650/120=5.4A Answer B • 8 Electrons flow counterclockwise in the picture so the greatest current will be where the electrons have not moved through resistors or at #1 Answer =A V I R P I V
Goal 4 • 9. In a series circuit there is only one path for electrons to flow in so if one bulb goes out they all go out. Parallel circuits have more than one path. Answer is D • 10. Answer A • 11. Parallel circuits have more than one path for electrons to flow Answer B • 12. P=IV I=2A V=12V 2 x 12 = 24W • 13. Series circuit more resistors dimmer the bulbs. This is not true in parallel circuits where the electrons have direct flow to each bulb. Answer=B
Goal 4 • 14. V = IR R=100Ω V=120V 120/100=1.2A Answer=B • 15. Iron filings take on the shape of the magnetic field that surrounds the magnet. The flow of the field is always out of the north pole of the magnet and into the south pole. Answer=D • 16. Moving the magnet in the wire coil causes the needle of the galvanometer to move. Answer=D • 17. The domains of the magnet remain aligned so you get two magnets. If the domains of the magnet move out of alignment the magnet loses its magnetism. Answer is C.
Goal 4 • 18. The more coils on an electromagnet the stronger it is picking up more paperclips. Answer is C. • 19. Increasing the current in an electromagnet increases its strength. The correct graph is B. • 20. Domains must be aligned in a permanent magnet. Answer B.
Goal 5 • 1. Dalton’s Theory: Elements are composed of the same kind of atom. Atoms of different elements are different. Compounds are made up of two or more elements. The atom was a solid indivisible sphere. The last was proven untrue when Thomson discovered electrons using the cathode ray tube to find the plum pudding model. Answer B • 2. Plum pudding model electrons embedded in proton sphere. Answer D • 3. Rutherford’s gold foil experiment determined the atom to be a dense positively charged nucleus with the electrons outside in an area of mostly empty space. Answer A.
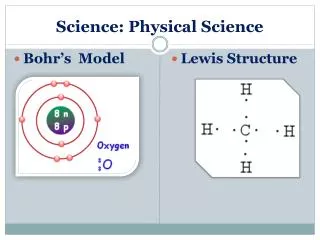
Goal 5 • 4. Bohr nucleus with electrons in specific orbitals or energy levels. Answer C. Be able to draw these. • 5. Current model- electron cloud model nucleus with protons and neutrons with electrons in an area of probability called a cloud. Answer C • 6. Similar properties is same family. Calcium is an alkaline earth metal with 2 valence electrons. A is oxygen with 6 valence electrons, B is carbon with four valence electrons, C is neon with 8 valence electrons, D is beryllium with 2 valence electrons. Answer is D
Goal 5 • 7. Atoms are arranged on the periodic table by the atomic number which is the number of protons the atom contains. Answer A • 8. Bromine is a halogen with 7 valence electrons and an oxidation # of -1. It is element 35 which is its number of protons so it has 35 protons and electrons. To determine the neutrons subtract 35 from the atomic mass. Answer D • 9. 121-51=70 Answer B • 10. To be a neutral atom the number of protons, positively charged subatomic particles must equal the number of electrons or negatively charged particles. Answer =D
Goal 5 • 11. Mass number = protons + neutrons. 14+16=30amu (atomic mass units). Answer=C • 12. Carbon-14 is an isotope of Carbon -12. 614C and 612C have an atomic number of 6 which means they are both carbon. The difference is the number of neutrons which shows up in the mass number of 14 and 12. Carbon-14 has 8 neutrons and Carbon-12 has 6 neutrons. Answer is A • 13. Isotopes have the same atomic number but different mass numbers. Atomic masses on the periodic table are the average of the isotopes found in nature. Answer A
Goal 5 • 14. Answer D see 12 for explanation • 15. Because chlorine has a mass of 35.45 that means there is very little chlorine 37 or the average of the isotopes would be closer to 37. Answer is A • 16. Density is a physical property of a compound. D=m/v m=3.8g. V=13-10ml 3.8/3ml=1.27g/cm³ Answer is D • 17. Aluminum’s volume of 1g is 1/2.7 or 0.37ml Iron’s volume of 1g is 1/7.9 or 0.13ml Lead’s volume of 1g is 1/11.4 or 0.09ml Silver’s volume of 1g is 1/10.5 or 0.1ml Answer is A. M D V
Goal 5 • 18. The most dense material is at the bottom of the container and the least dense is at the top. Anything with a density less than 1.0g/ml will float on water. Anything more will sink. Answer D • 19. Nitrogen, Oxygen, and Hydrogen will become liquids above their melting points which -200ºC is. Chlorine is still a solid because the temperature has not warmed up to -101ºC. Answer is A. • 20. Heat of fusion where solid becomes liquid is -10ºC. Heat of vaporization where liquid becomes a gas is 90ºC. Answer is C
Goal 6 • 1. Calcium and potassium are not in the same family so they do not have the same # of valence electrons or oxidation number. Only the same element have the same #of protons. Answer is D. They both are in the 4th energy level or row. • 2. Far left sides of the periodic table have positive oxidation numbers meaning that they want to lose electrons. They also are metals. Answer is D. • 3. Group 2A or 2 are the Alkaline Earth Metals. They have an oxidation number of +2 therefore they will react with something that has a negative oxidation #. • Alkali metals +1, Halogens-1, noble gases stable, transtion metals change positive oxidation states. Answer is B.
Goal 6 • 4. Period is same row. Silver and Tin are in Period 5. Answer is C • 5. Al+3 F-1 crisscross Answer is C • 6. Neon is stable getting rid of A. Lithium, Beryllium and Boron all have +oxidation numbers getting rid of C and D. Answer is B because Lithium is a +oxidation # and Fluorine is a –oxidation #. • 7. Covalent bonds occur between 2 nonmetals and electrons are shared. Potassium and Calcium are both metals. Answer is A. • 8. A is a synthesis reaction • B is a double replacement reaction • C is breaking water apart into smaller substances so it is the decomposition • D is a synthesis reaction Answer is C
Goal 6 • 9. A is a decomposition reaction • B is a double replacement reaction • C is a synthesis reaction • D is a single replacement reaction. Answer is B. • B is not a neutralization reaction where and acid and a base form salt and water. • 10. Reactants are on the right, products are on the left of the equation. Reactant is Fe2O3. Products are 4Fe and 3O2. Answer is C. This is a decomposition reaction
Goal 6 • 11. A. Reactants 2K, 1C, 3Om 1Ba, 2Cl • Products 1K, 1Cl, 1Ba, 1C, and 3O • B. Reactants2K, 1C, 3O, 1Ba, 2Cl • Products 2K, 2Cl, 1Ba, 1C, 3O • C. Reactant 2K, 1C, 3O, 2Ba, 4Cl • Products 2K, 2Cl, 1Ba, 1C, 3O • D. Reactants 2K, 1C, 3O, 2Ba, 4Cl • Products 1K, 1Cl, 2Ba, 2C, 6O • Answer is B
Goal 6 • 12. Exothermic means heat is a product of the chemical reaction (heat out). Answer is A. This is a sign of a chemical change • 13. A solid that falls out when 2 liquids are poured together is a precipitate. It is a sign of a chemical change. Answer is A • 14. The one that is the lowest slope has the least response to a temperature change. The answer is KCl. HCl and NH3 are both gases because their solubility decreases as they warm up. • 15. 0-6.9 acid, 7.1-14 base, 7 neutral. Answer is A
Goal 6 • 16. As you move away from 7 the acid or base gets stronger. Cola is the strongest acid and Bleach is the strongest base. Answer is D. • 17. Solids are more soluble when you crush increasing the surface area, when you heat it making the molecules move faster, and when you stir it moving the molecules more. Answer is A. Gases go into solution easier under pressure and cold. • 18. Ionic bonds conduct electricity. Salt is an ionic bond. Cornstarch, sugar, and oil are all covalent.
Goal 6 • 19. Alpha particles are 24He. When one is subtracted from an element the mass number decreases by 4 and the atomic number decreases by 2. A Beta particle is 10e. To get a Beta particle released a neutron is converted to a proton and a electron. The mass number does not change because an electron is removed. The atomic mass increases because a proton is gained. A gamma particle does not cause a change in mass or atomic number because it has no mass and is made up of photons. Answer is A • 20. Fission is splitting of a nucleus into two smaller nuclei and fusion is the combining of two smaller nuclei into 1 larger nucleus. Answer is B.