Covalent Bonds
Covalent Bonds. http://www.ptable.com. Octet Rule must be satisfied-reach noble gas state What of elements that meet and cannot completely transfer e- and satisfy octet rule for each? H and H atom story…one happy if e- tranfer other sad. Both atoms get one e-

Covalent Bonds
E N D
Presentation Transcript
http://www.ptable.com • Octet Rule must be satisfied-reach noble gas state • What of elements that meet and cannot completely transfer e- and satisfy octet rule for each? • H and H atom story…one happy if e- tranfer other sad
Both atoms get one e- • Both satisfy octet rule by SHARING • Each atom must have both e- • DIRECTIONAL bond- product of shared e- • Each H atom will have full outer energy level
Hydrogen gas H2 • Two H atoms sharing each other’s e-
Oxygen gas shares 4 valence e- or 2 pairs valence e- • Results in a double bond • Nitrogen gas shares 3 pairs of valence e- • Results in triple bond • Hydrogen, fluorine, nitrogen, chlorine… • BrINClOF (Chlorine is capital C lower case l)
Remember how to do this?1s2… • Each bond represents 2 shared e-
Nature of Covalent Bonds • Strong electrostatic attraction between shared valence e- and + nucleus • One end of molecule has more + than – charge
Draw Lewis dots and show how water molecule shares e- • Covalent bond VERY STRONG • Shared e- confined so bond is DIRECTIONAL
Covalent vs. Ionic Bonds • http://www.mhhe.com/physsci/chemistry/animations/chang_7e_esp/bom1s2_11.swf
http://www.bing.com/videos/search?q=ionic+vs+covalent+bonding&FORM=VIRE3&adlt=strict#view=detail&mid=D0981F9E5http://www.bing.com/videos/search?q=ionic+vs+covalent+bonding&FORM=VIRE3&adlt=strict#view=detail&mid=D0981F9E5 • video
H tends to share only e- Octet rule not satisfied by giving up e- • H and Cl… HCl • Chlorine tends to grab at e- • Doesn’t want to give it up… • Uneven distribution of e- density • ELECTRONEGATIVITY becomes important ᵟsmall delta indicates charge – or + ᵟ+ H Cl ᵟ-
Result is… • POLARITY • A partial charge separation • Electronegativity increases e- moves towards the area • In covalent bonds, neither atom has energy to remove e- from other atom
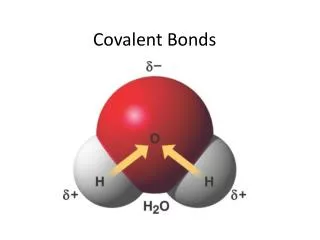
Example of a polar covalent molecule • H-O-H Water H2O • Stable octet for oxygen.. Completed with other H atom
Notice how directional the bonds are for water- a covalent molecule • Polarity of water makes it the universal solvent.. Able to dissolve many substances.. • Not all!!
Non Polar Molecules • Even distribution of charge • Examples: oxygen gas O2
Cov vs. Ionic Bonds Electronegativity counts • Hydrogen Gas molecule: H2 No difference of attraction of electrons between 2 atoms so ΔEN=O (Capital Delta Δ means change) Hydrogen gas is a non polar covalent molecule Water is polar covalent
EN and bond types • 0 = non polar cov • < 0.3 no polar cov • 0.3- 0.4 slightly polar cov • 1.0-1.7 polar cov • More and more separation of charges • If difference > 1.7 then generally ionic!
Go to ptable.com • http://www.ptable.com
Your Turn • Use EN # on PT to determine difference in EN between elements of a compound and type of bond formed between them. NaBr Water CS2
Inter and Intra prefixes • Forces may be intra or intermolecular in nature. • H:H molecule of hydrogen gas • Can 3rd H come into the molecule? • No Pauli Exclusion Principle • Molecules formed with hydrogen gas. • Bond very strong .. What’s in between molecules?
BLACK Intra Forces-within molecule-RED Inter Forces- between or among molecules as they exist in natural state Intermolecular forces weaker than intramolecular forces The unit formed is called a molecule
Properties of Covalent (diatomic) Elements and Compounds • Dull no free e- • Soft, brittle- weak intermolecular forces • Low mp and bp- weak intermolecular forces • NO conductivity as solid or molten (no charges) • Some soluble in water Aqueous solutions DO NOT conduct electricity at all- no free moving charged particles
Diamond and graphite examples of Carbon in covalent bonding • Microstructures in diamond very different from graphite • Both covalent, both made of carbon only • Diamond- strong interactions of directionality of carbon atoms- lattice like STRONG BONDS • Graphite- weaker bonds due to direction and spaces between atoms allows for breakage- leaving of powder from pencil
Molecular Compounds Nomenclature • State how many of each element in compound • Water H-O-H • FIRST: must know prefixes: • 1. Mono • Di • Tri • Tetra • Penta • Hexa • Hepta • Octa • Nona • Deca
Communicate first element • Assume same name as element’s name • 2nd element “ide” notation • ONE exception: if 1 is coefficient for 1st element then it is dropped • WATER? Dihydrogen monoxide
Your Turn • N2O5 • Carbon dioxide.. To write this formula properly, what is dropped? • Sulfur trioxide • SF6