Energy levels and sub-levels
230 likes | 785 Views
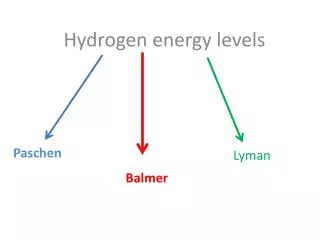
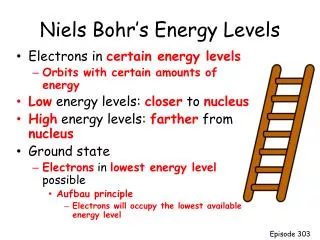
Energy levels and sub-levels. Electron Configurations. Energy levels and sub-levels. Energy levels. These are broadly similar to the “shells” used in the Bohr Model.

Energy levels and sub-levels
E N D
Presentation Transcript
Energy levels and sub-levels Electron Configurations
Energy levels and sub-levels Energy levels • These are broadly similar to the “shells” used in the Bohr Model • There are 4 main energy levels. The main energy levels are called the Principal Quantum numbers and abbreviated with the letter “n”. • Each main energy level has a corresponding number of sub-levels. So the first energy level has one sub-level, the 2nd level has two sub-levels, the 3rd has three, and so on.
Energy levels and sub-levels Sub-levels The main energy levels contain sub-levels The different main energy levels have different numbers of sub-levels in them There are four types: s, p, d, f
Energy levels and sub-levels: Orientation • The sub-levels themselves may have different orientations where the electrons can reside. • s has 1 orientation • p has 3 orientations • d has 5 orientations • f has 7 orientations
Energy levels and sub-levels: Spin • Each level, sub-level, orientation can have a maximum of 2 electrons. • Since the electrons are existing in the same “energy-space”, they must avoid interfering with each other. (They have similar charges, and so should repel each other) • To avoid this, they must be “spinning” in opposite directions. This is often refered to as “spin-up and “spin-down”.
s 1 2 Energy levels and sub-levels: Levels 1 and 2 s 1 1 2 2 8 2 p 3 6
p 3 6 s 1 2 Energy levels and sub-levels: Level 3 18 d 10 5
Energy levels and sub-levels: extended There are three more main levels after Level 4 Their energy sub-levels and orientations are described with the same rules that apply to Level 4 So Level 5 has 5s. 5p, 5d, and 5f sub-levels Level 6 has 6s, 6p, 6d and so on sub-levels
Credits • With the inspiration and original work of Nigel Saunders, Creative Chemistry