Nuclear Energy
Nuclear Energy “Energy will be so cheap, it won’t even be metered…” Atomic Structure Nucleus - consists of protons and neutrons; contains almost all of the mass of the atom; held together by strong and weak nuclear forces Protons - positively charged particles; mass = 1 AMU

Nuclear Energy
E N D
Presentation Transcript
Nuclear Energy “Energy will be so cheap, it won’t even be metered…”
Atomic Structure Nucleus - consists of protons and neutrons; contains almost all of the mass of the atom; held together by strong and weak nuclear forces Protons - positively charged particles; mass = 1 AMU Neutrons- neutrally charged particle; mass = 1 AMU Electrons - negatively charged particle; mass = .0006 AMU
Electrical Structure The energy of an electron in an atom is quantized, i.e. it can only have discreet values In order to change energy from one level to another, an electron must absorb or emit a quantum of energy equal to the difference in the energy levels This quantum of energy will be in the form of electromagnetic radiation. The frequency of the radiation will be given by the equation E = hf, where h = Planck’s constant
Nuclear Structure The nucleus is composed of protons and neutrons. The positive charge of the protons causes them to be repelled. However, the strong and weak nuclear forces overcome this repulsion. The type of element that an atom is depends upon the number of protons in the nucleus. For a given number of protons, there are many different numbers of neutrons that are allowed in the nucleus.
Ions Versus Isotopes Ions are atoms with differing numbers of electrons and protons.These atoms have a net charge, and are very chemically reactive. Ex. Ca+1 is a calcium atom that has one less electron than protons Isotopes of an element are atoms that have differing numbers of neutrons in the nucleus. They are all chemically the same.Ex. Uranium-235 has 92 protons and 143 neutrons; uranium-236 has 92 protons and 144 neutrons
Designation Isotopes are designated by the total number of protons and neutrons in the atom. This number is usually posted to the upper left of the chemical symbol. Ex.: 14C = carbon-14 The way to figure out the number of neutrons in an atom is to subtract the number of protons from this number. Ex.: carbon has 6 protons (check atomic chart)carbon-14 has 14 - 6 = 8 neutrons
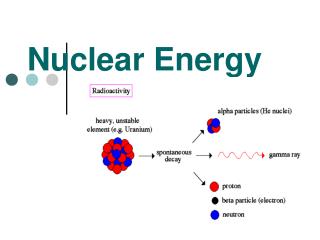
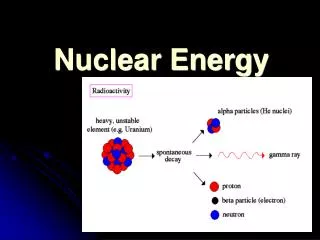
Radioactivity Not all isotopes of an atom are stable. Some elements have no stable isotopes. If it is unstable, it will decay alpha decay - emission of an alpha particle (2 protons + 2 neutrons) beta decay - emission of a beta particle (electron or positron) gamma decay - emission of electromagnetic radiation
Exponential Decay Experimentally, we know that radioactive materials decay exponentially, i.e. the same percentage decays in the same amount of time Half-life - the amount of time that it takes for half of a substance to decay Activity - how much material is decaying per unit time; it is inversely proportional to the half life (longer the half-life, the less the activity)
Half-life Example Iodine-131 has a half life of 8 days. If I start with 10 kg. of it, how much do I have after 24 days? 24 days/(8 days/half-life) = 3 half-lives 10 kg/2 = 5 kg 5 kg/2 = 2.5 kg 2.5 kg/2 = 1.25 kg
Binding Energy When radioactive decay occurs, energy is released. From where does it come? Binding energy - the amount of energy holding the nucleus together; the lower the binding energy, weaker the nucleus is held together
Fission Versus Fusion If a nucleus can become more tightly bound, it will. It can do this one of two ways. Fusion - nuclei come together Fission - nucleus breaks apart When either of these occurs, energy is released.
Fusion If all of the elements below iron-56 can become more tightly bound by fusing, then why do they not do it spontaneously? Answer: In order to fuse, the nuclei must overcome the electronic repulsion of the protons. The strong and weak nuclear force are very short range forces, i.e. they operate on a scale of 10-15 m. Nuclei must get close for them to pull the nuclei together.
Using This Energy If energy is released each time fusion or fission occurs, then we should be able to absorb this energy and use it. Problem with fusion: We have not been able to replicate the conditions necessary (high temperature and pressure) in a controlled way. We have done the uncontrolled method.Ex.: hydrogen bomb Problem with fission: For a viable reactor, you are going to need a lot of energy released in a short period of time. This means that you need a radioactive substance with a high activity. Where are you going to find this? Answer: No where in nature since elements with short half lives decayed away a long time ago.
Catalysts We can convert non-radioactive or long half-life radioactive atoms into short half-life radioactive atoms. The process to do this is neutron bombardment Ex.: Calcium-40 captures a neutron; it becomes unstable and decays via gamma decay
Practicality We can cause any atom to become radioactive. However, if we are having to put energy into the system, this limits the net amount that we can get out. Question: Can we find a natural source of neutrons? Answer: Yes, some isotopes will produce neutrons, which will provide the catalyst to keep the reaction going
Chain Reaction Uranium-235 is the one natural isotope that is abundant enough for use in a commercial reactor U235 + n -> U236 -> 2 new isotopes + energy + 2n The two neutrons that are given off by the reaction can be used to cause 2 other uranium-235 isotopes to decay.
Controlling the Reaction This ability of uranium to create the catalyst that keeps the reaction going allows for a sustained chain reaction. However, to keep the reaction going and to keep it from getting out of control, you need: 1) Neutron moderator - the neutron that U235 absorbs best is a slow moving neutron; this means that something has to slow down those produced in the reaction 2) Neutron absorber - since the reaction produces 2 or more neutrons, some neutrons will have to be absorbed, or the rate of the reactions will increase exponentially
American Nuclear Reactor Water acts as a neutron moderator and a heat transfer medium Absorb excess neutrons Steam from here is not radioactive
Safety Features Most of the world uses a design similar to the one on the previous slide. It has several safety features to contain the radioactivity. 1) If a water leak develops in the reaction chamber, then the temperature will increase because heat is not being removed. However, the reaction will slow down since moderator is gone. 2) In the event of an electrical problem, the control rods fall into the reaction chamber and absorb all neutrons, shutting reactor down 3) Reaction chamber has enough concrete and steel to take a hit from a 747 aircraft
Accidents Chernobyl - bad technicians working with a bad design; Soviet RMBK design reactor uses water only as a heat transfer fluid; helium and carbon are the neutron moderators; technicians were running unauthorized test to see how many safety features could be turned off before trouble occurred; they found out Three Mile Island - technician tied valve shut while doing maintenance; when temperature got to high, computer was not able to open the valve to cool things off; top of reactor partially melted; small amount of radioactive steam was vented to outside; radioactivity released almost undetectable, but panic ensued
Where We Stand There are over 110 operating reactors in the U.S. Most are in the East. The last new reactor was finished in the late 1990’s; it took 23 years to build it. Even though nuclear reactors have proven to be very safe in the U.S., the industry is, essentially, on its way out. There are no plans for any new reactors to be built. However, this does not mean that we can forget about nuclear energy. We still have the issue of waste with which to contend.
Nuclear Fuel Cycle • Mining - extracted as uranium oxide; most in the Western U.S.tilling piles and ponds contain heavy metals and radioactivity • Enrichment - need to increase the concentration of U235 for usein the reactor • Fuel Rod - concentrated uranium is made into pellets, packedinto a rod, and put in the reactor • Fuel Reprocessing - after some time, the amount of usable fuelin the rod is too low; re-process rod to remove usable fuelfor use in a new rod • Disposal - all non usable fuel and waste will need disposal;currently, there is no facility for this in the U.S.
Current Status of Waste Currently, all high level nuclear waste is stored onsite in either pools of water or in above ground barrels The U.S. government was supposed to have completed a waste repository by 1998 that would take all of this waste. Lawsuits and studies have delayed this. The situation is getting critical at some locations. A temporary solution is being sought. However, no state wants the waste. Possible solution: temporary storage on Native American reservations since they do not have to follow state laws.