Molecular Structures
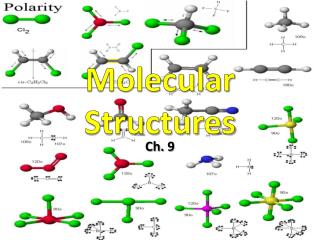
Molecular Structures. Chapter 9. Molecular Models. Molecular geometry is the general shape of a molecule, as determined by the relative positions of the atomic nuclei. We use molecular models to visualize and understand shapes of molecules. Molecular Structures.

Molecular Structures
E N D
Presentation Transcript
Molecular Structures Chapter 9
Molecular Models Molecular geometry is the general shape of a molecule, as determined by the relative positions of the atomic nuclei. We use molecular models to visualize and understand shapes of molecules
The Valence-Shell Electron Pair Repulsion Model The valence-shell electron pair repulsion (VSEPR) model predicts the shapes of molecules and ions by assuming that the valence shell electron pairs are arranged as far from one another as possible to minimize the repulsion between them. • To predict the relative positions of atoms around a given atom using the VSEPR model, you first note the arrangement of the electron pairs around that central atom.
The Valence-Shell Electron Pair Repulsion ModelElectron Pair Geometry vs Molecular Geometry Electron Pair Geometry – is determined by the number and arrangement of all electron pairs (bonding and lone pairs) around the central atom. Molecular geometry – is determined by the arrangement of atoms in space. In molecules with no lone pairs Electron Pair Geometry and Molecular geometry are the same.
Predicting Molecular Geometry • Draw the Lewis structure • Determine how many electron pairs are around the central atom. Count a multiple bond as one pair. • Decide on the best (minimized repulsion) arrangement of bonds and lone pairs around the central atom • Decide on electron pair geometry. • If molecule has no lone pairs around the central atom you are done. • If the molecule has lone pairs around the central atom – decide on molecular geometry
Why is tetrahedral shape preferred over square planar in case of 4 bonds around the central atom? X 90º X – A – X X
Predicting Molecular Geometry Two electron pairs –BeCl2 Cl Be Cl linear arrangement. AX2 • The electron pair geometry and the molecular geometry • of beryllium dichloride are linear. Bond angle is 180o.
Predicting Molecular Geometry trigonal planar arrangement. .. Three electron pairs – boron trifluoride BF3 :F: B .. .. :F F: .. .. AX3 The electron pair geometry and the molecular geometry of BF3 is trigonal planar.
: : : S : : :O O Predicting Molecular Geometry trigonal planar arrangement. Three electron pairs – SO2 AX2E1 • The electron pair geometry is trigonal planar. • Since one of the electron pairs is a lone pair, the molecular • geometry is described as bent or angular.
H Predicting Molecular Geometry tetrahedral arrangement. Four electron pairs – methane CH4 H C H H AX4 The electron pair geometry and the molecular geometry of CH4 is tetrahedral.
H Predicting Molecular Geometry tetrahedral arrangement. Four electron pairs – ammonia NH3 .. N H H AX3E1 • The electron pair geometry is tetrahedral Since one of the electron pairs is a lone pair, the molecular geometry is described as triangular pyramidal.
Predicting Molecular Geometry tetrahedral arrangement. Four electron pairs – water H2O .. :O H H AX2E2 • The electron pair geometry is tetrahedral Since two of the electron pairs are lone pairs, the molecular geometry is described as bent
Predicting Molecular Geometry Four electron pairs -tetrahedral arrangement. Order of increasing repulsion: bonding pair – bonding pair < bonding pair – lone pair < lone pair – lone pair
Example Describe the molecular geometry in boron trihydride, BH3 and phosphorus trihydride, PH3 : : :F: :F: : : : : :F B F: :F P F: : : : : :
: : : F : F : : F : : : : : F : P : F : : Predicting Molecular Geometry trigonal bipyramidal arrangement. Five electron pairs - phosphorus pentafluoride, PF5 AX5 The electron pair geometry and the molecular geometry of PF5 is trigonal bypiramidal
F F : S F F Predicting Molecular Geometry trigonal bipyramidal arrangement. Five electron pairs – sulfur tetrafluoride, SF4 AX4E1 • The electron pair geometry is trigonal bipyramidal Since one of the electron pairs is a lone pair, the molecular geometry is described as sea-saw
F : F Br : F Predicting Molecular Geometry trigonal bipyramidal arrangement. Five electron pairs – bromine trifluoride, BrF3 AX3E2 • The electron pair geometry is trigonal bipyramidal Since two of the electron pairs are lone pairs, the molecular geometry is described as T shaped
Predicting Molecular Geometry trigonal bipyramidal arrangement. Five electron pairs – xenon difluoride, XeF2 F : : Xe : F AX2E3 • The electron pair geometry is trigonal bipyramidal Since three of the electron pairs are lone pairs, the molecular geometry is described as linear
: : : : : F: F: :F :F: : : : : Predicting Molecular Geometry octahedral arrangement Six electron pairs – sulfur hexafluoride, SF6 S :F :F: : AX6 The electron pair geometry and the molecular geometry of SF6 is octahedral
F F F I F F : Predicting Molecular Geometry octahedral arrangement Six electron pairs – iodine pentafluoride, IF5 AX5E1 The electron pair geometry is octahedral Since one of the electron pairs is a lone pair, the molecular geometry is described as square pyramidal
Predicting Molecular Geometry octahedral arrangement Six electron pairs – xenon tetrafluoride, XeF4 : F F Xe F F : AX4E2 The electron pair geometry is octahedral Since two of the electron pairs are lone pairs, the molecular geometry is described as square planar
Valence Bond Theory Valence bond theory is an approximate theory to explain the covalent bond from a quantum mechanical view. • According to this theory, a bond forms between two atoms when the following conditions are met. • Two atomic orbitals “overlap” to form a molecular orbital. • The total number of electrons in both orbitals is no more than two.
H 1s1 H 1s1 F[He]2s22p5 1s 2s 2px 2py 2pz F[He]2s22p5 2s 2px 2py F[He]2s22p5 2s 2px 2py
However, carbon, with only two unpaired electrons, generally forms four bonds. For example, methane, CH4, is well known. Hybrid Orbitals We might expect the number of bonds formed by an atom would equal its unpaired electrons. • Chlorine, for example, generally forms one bond and has one unpaired electron. • Oxygen, with two unpaired electrons, usually forms two bonds.
2s 1s 1s 2p 2p 2s Energy C atom (ground state) C atom (promoted)
2p sp3 sp3 2s Energy 1s 1s Hybridization of carbon in CH4 C-H bonds 1s C atom (promoted) C atom (hybridized state) C atom (in CH4)
Hybrid Orbitals Hybrid orbitals are orbitals used to describe bonding that are obtained by taking combinations of atomic orbitals of an isolated atom. • In case of CH4, a set of hybrids is constructed from one “s” orbital and three “p” orbitals • sp3 hybrid orbitals. • The four sp3 hybrid orbitals take the shape of a tetrahedron
2p 2s Energy 1s 1s Hybridization of nitrogen in NH3 sp3 sp3 lone pair lone pair N-H bonds 1s N atom N atom (hybridized state) N atom (in NH3)
H : H O : Example Describe the bonding in H2O according to valence bond theory. Assume that the molecular geometry is the same as given by the VSEPR model. • From the Lewis formula for a molecule, determine its geometry about the central atom using the VSEPR model. The molecular geometry is tetrahedral
2p sp3 sp3 2s Energy 1s 1s Hybridization in H2O lone pairs O-H bonds 1s O atom (ground state) O atom (hybridized state) O atom (in H2O)
2s 1s 1s Hybridization of boron in BH3 2p 2p 2s Energy B atom (ground state) B atom (promoted)
Energy Hybridization of boron in BH3 2p sp2 B-H bonds 2s 1s B atom (promoted) B atom (hybridized state) B atom (in BH3)
2s 1s 1s Hybridization of beryllium in BeH2 2p 2p 2s Energy Be atom (ground state) Be atom (promoted)
Energy Hybridization of beryllium in BeH2 2p sp B-H bonds 2s 1s Be atom (promoted) Be atom (hybridized state) Be atom (in BeH2)
Cl Cl Cl P Cl Cl Describe bonding in PCl5 using hybrid orbitals. • The Lewis formula of PCl5 is
3d 3p 3s P atom (ground state)
3d 3p 3s P atom (promoted)
3d sp3d P atom (hybridized state)