CHAPTER 24 Nuclear Energy
80 likes | 325 Views
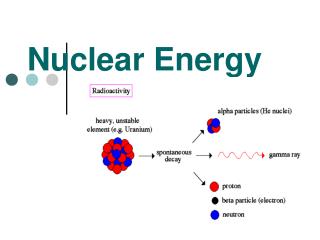
CHAPTER 24 Nuclear Energy. I. Radioactivity (p.674-683). A. Definitions. Radioactivity emission of high-energy radiation from the nucleus of an atom Nuclide nucleus of an isotope Transmutation process of changing one element into another via nuclear decay. B. Types of Radiation.

CHAPTER 24 Nuclear Energy
E N D
Presentation Transcript
CHAPTER24 Nuclear Energy I. Radioactivity (p.674-683)
A. Definitions • Radioactivity • emission of high-energy radiation from the nucleus of an atom • Nuclide • nucleus of an isotope • Transmutation • process of changing one element into another via nuclear decay
B. Types of Radiation • Alpha () • helium nucleus paper 2+ • Beta-minus (-) • electron lead 1- • Gamma () • high-energy photon concrete 0
C. Nuclear Decay • Why nuclides decay… • to obtain a stable ratio of neutrons to protons Stable Unstable (radioactive)
C. Nuclear Decay TRANSMUTATION • Alpha Emission • Beta Emission
Example Half-lives polonium-194 0.7 seconds lead-212 10.6 hours iodine-131 8.04 days carbon-14 5,370 years uranium-238 4.5 billion years D. Half-life • Half-life (t½) • time it takes for half of the nuclides in a sample to decay
D. Half-life • How much of a 20-g sample of sodium-24 would remain after decaying for 30 hours? Sodium-24 has a half-life of 15 hours. GIVEN: total time = 30 hours t1/2 = 15 hours original mass = 20 g WORK: number of half-lives = 2 20 g ÷ 2 = 10 g (1 half-life) 10 g ÷ 2 = 5 g (2 half-lives) 5 g of 24Na would remain.