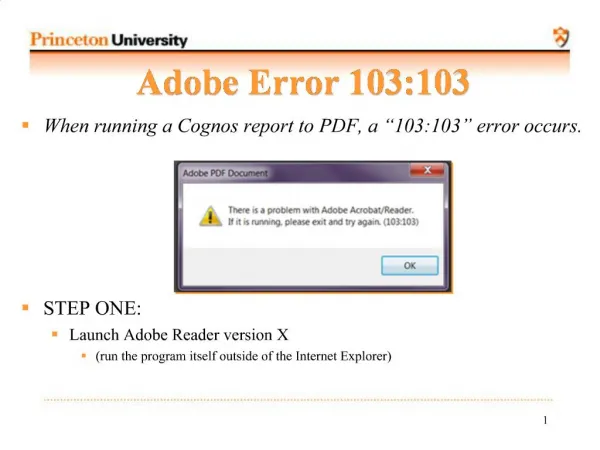
Understanding Radioactive Decay: Half-Life, Decay Constant, and Exponential Behavior
This review of Dr. Cecilia Vogel's lecture explores the fundamentals of nuclear physics, focusing on key concepts such as radioactive decay, half-life, and decay constants. Students will learn about the probability of nuclear decay, how to measure decay rates, and the significance of exponential decay in nuclear processes. The lecture covers practical applications, including how to determine decay from half-lives, as well as how to interpret data from isotopes. Detailed examples illustrate these concepts, making complex ideas accessible.

Understanding Radioactive Decay: Half-Life, Decay Constant, and Exponential Behavior
E N D
Presentation Transcript
PH 103 Dr. Cecilia Vogel Lecture 23
Review • Nuclei • a, b ,g decays • Radiation damage Outline • Nuclear physics • exponential decay • decay constant • half-life
How Quickly Decay Occurs • As with all quantum processes it’s all probability. • Cannot determine when a particular nucleus will decay, • just probability that it will have decayed in a certain time. • Can predict how many will decay in certain time, but not which ones. • for example can predict how long before half have decayed = half-life
Half-Life • Half-life is • the time it takes for the number of radioactive nuclei to decrease by half. • After another half-life, half of what’s left will be gone. • Now only 1/4 left. • After another half-life, half of what’s left will be gone. • Now only 1/8 left. • etc • This kind of behavior is • exponential decay.
Half-Life • This kind of behavior is exponential decay: • N = # nuclei left at time t • No = initial # nuclei • l = decay constant • (not wavelength!)
Decay Constant • Decay Constant, l • how fast it decays • higher l, faster decay • inversely proportional to half-life
Where to Find the Info • Appendix B, of course! • Find the isotope you’re interested in, • rightmost column holds half-lives. • If there’s nothing in that column, • it’s not radioactive. • BEWAREof units – they vary! • From the half-life, you can find the decay constant, • from the decay constant, you get decay as function of time.
Working Backward • If you know the decay constant and the time, you can plug into exponential. • What if you need to calculate the decay constant or the time from N’s? • Solving decay eqn yields:
Decay Rate • Another quantity of interest is • The number of nuclei that decay per unit time, called the decay rate • For each nucleus that decays • one emitted particle will be counted. • Decay rate = activity • Decay rate is also an exponential function of time.
Units • Units of t and t½ are time units: • seconds, minutes, years, etc. • Units of l are inverse time • Be consistent with your time units!! • N and No are unitless numbers • or moles • Units of R and Ro • 1 decay/s = 1 Bq = 1 Bequerel (sometimes written as s-1) • 1 Ci = 1 Curie = 3.7X1010 Bq