Understanding Chemical Equilibrium in Reactions
Learn why reactions proceed in specific directions, the role of entropy, equilibrium constants, reaction rates, Le Chatelier’s Principle, and factors affecting equilibrium. Understand key concepts with examples.

Understanding Chemical Equilibrium in Reactions
E N D
Presentation Transcript
Chemical Equilibrium PHYS 1090 Unit 15
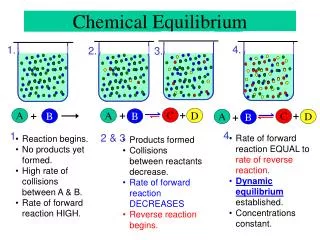
Direction • A chemical reaction proceeds in a specific direction under specific conditions. A B • Why? Entropy increases!
Increase Entropy • Disperse matter • Dissolve a solid in a liquid • Melt a solid • Release a gas • Disperse energy • Generate heat • Biggest effect at low temperature • When they conflict, one dominates. • Direction can change with temperature.
Uncomplicated Examples • Dissolve CaCl2 in water • Gets hot: disperses energy • Solid dissolves: disperses matter • Burn wood: (C6H10O5)n(s) + 6n O2(g) 6nCO2(g) + 5nH2O(g) • Gets hot: disperses energy • Generates CO2 and H2O gas: disperses matter
Complicated Examples • Dissolve NH4NO3 in water • Gets cold: constrains energy • Solid dissolves: disperses matter • Here, dispersing matter wins • Solidify super-cooled sodium acetate • Gets hot: disperses energy • Liquid solidifies: constrains matter • Here, dispersing energy wins
Reaction Rates A B • Rate is D[B]/Dt= –D[A]/Dt • Depends on • Intrinsic reactivity of A • Temperature • How much A there is • Proportional to [A]
Reaction Rates A+ B C • Rate is D[C]/Dt= –D[A]/Dt =–D[B]/Dt • Depends on • Intrinsic reactivity of A and B • Temperature • How much A and B there are • Proportional to [A][B]
Rate constants A + B C • Rate = k[A][B] • k = rate constant • Independent of [A], [B] • Depends on temperature
Equilibrium A B • Two reactions: A B and B A • If both have the same rate, [A] and [B] don’t change • “Equilibrium” • kf[A] =kb[B] • [B]/[A] = kf /kb = K= equilibrium constant
Equilibrium Constants • For A + B C • For A + A B • For A + B C + D
K = [HCl]2 K = [H2][Cl2] Examples NH3 + HCl NH4Cl [NH4Cl] [NH3][HCl] H2 + Cl2 2 HCl
Unchanging Concentrations • Solvent • Solid • Unmixed liquid Drop out of equilibrium constant
[H2O][NaCl] K = [KOH][HCl] [NaCl] K = [KOH][HCl] Example KOH(aq) + HCl(aq) H2O + NaCl(aq) • But H2O is the solvent: its concentration does not change. So:
[H+][OH–] K = [H2O] Another Example H2O H+(aq) + OH–(aq) • But again H2O is the solvent. So: K = [H+][OH–]
[CaCO3][NaCl]2 K = [CaCl2][Na2CO3] [NaCl]2 K = [CaCl2][Na2CO3] Precipitation Example CaCl2(aq) + Na2CO3(aq) CaCO3(s) + 2 NaCl (aq) • CaCO3 is a solid precipitate, not dissolved. So:
Le Chatelier’s Principle • If a chemical system at equilibrium is perturbed, the system will adjust to minimize the perturbation
Instances • If reactant concentration is high: • More product will be generated • If products are removed: • More product will be generated • If products are insoluble: • Effectively removes them • More product will be generated • (precipitation reactions)
Instances • If a reaction generates a gas: • Increasing the pressure consumes products • Decreasing the pressure consumes reactants • Letting gas escape generates more • If a reaction generates heat: • Increasing the temperature inhibits it • Decreasing the temperature facilitates it