The Representative Elements
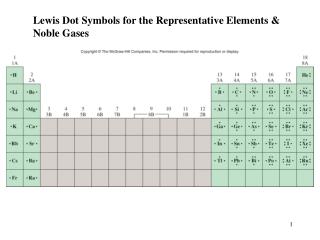
The Representative Elements. Ch 18 & 19. Representative Elements. Their chemical properties are determined by their valence (s and p) electrons. Properties are similar within a group, but first element in a group tends to act differently due to smaller size.

The Representative Elements
E N D
Presentation Transcript
The Representative Elements Ch 18 & 19
Representative Elements • Their chemical properties are determined by their valence (s and p) electrons. • Properties are similar within a group, but first element in a group tends to act differently due to smaller size. • Most abundant element is oxygen, followed by silicon. • Most abundant metals are aluminum and iron, which are found as ores.
Group 1 (1A) – Alkali Metals • Have Valence electron configuration ns1 • Will lose 1 electron to form M+ Ions • React vigorously with water to form M+ and OH- ions and hydrogen gas. • React with oxygen to form oxides. Lithium will form a regular oxide (Li2O) while sodium will form a peroxide (Na2O2). Potassium, rubidium and cesium form superoxides (MO2)
Hydrogen • Can form covalent compounds with other nonmetals. • Will form salts, hydrides, with very active metals (group 1A and 2A). • Hydride ion, H-, is a strong reducing agent. • Covalent hydrides form when hydrogen bonds with other nonmetals. • Metallic hydrides occur when hydrogen atoms migrate into transition metal crystals.
Group 2 (2A) – Alkaline Earth Metals • Have valence electron configuration ns2 • Called Alkaline earth because of the basicity of their oxides. • React less vigorously with water than group 1. • Heavier alkaline earth metals form ionic nitrides and hydrides. • Hard water is caused by the presence of Ca+2 and Mg+2 ions.
Group 13 (3A) • Have valence electron configuration ns2 np1 • Show increasing metallic character goingdown the group. • Boron forms covalent compounds with hydrides called boranes. These compounds are highly electron deficient and very reactive. • Aluminum has some covalent characteristics as do indium and gallium. • Thallium is completely metallic in character.
Group 14 (4A) • Have valence electron configuration ns2 np2 • Show a change from nonmetallic to metallic properties going down the group. • All elements in this group form covalent bonds with nonmetals. • MX4 compounds (except carbon)react with Lewis bases to form two additional covalent bonds.
Group 15 (5A) • Have varied chemical properties. • All members except nitrogen form molecules with 5 covalent bonds. (Nitrogen has no d sublevel) • Nitrogen and Phosphorous are nonmetals and form 3- anions in salts with active metals. • Antimony and bismuth are metallic. However their 5+ cations tend to be molecular rather than ionic.
Nitrogen • The strength of the triple bond in the N2 molecule is important both thermodynamically and kinetically as they decompose exothermically. • The nitrogen cycle is the process through which nitrogen is recycled through the environment. • Nitrogen forms a series of oxides in which it has an oxidation state ranging from 1 to 5. • Nitric acid is a strong acid which is important as a reducing agent. • Ammonia is the most important nitrogen hydride. • Has pyramidal molecules with polar bonds. • Hydrazine (N2H4) is a powerful reducing agent.
Phosphorous • Exists in three elemental forms: white, black and red. • Phosphine (PH3) has a structure analogous to that of ammonia but with bond angles closer to 90o. • Forms two oxides with oxidation states of 3+ and 5+.
Group 16 (6A) • Shows the usual tendency of increasing metallic properties going down the group. • None behave as typical metals • Achieve noble gas configurations by adding two electrons to form 2- anions. • Form covalent bonds with other nonmetals. • Oxygen exists in two elemental forms: O2 and O3. • Sulfur has two elemental forms, both of which contain stacks of S8 rings. • Sulfur also forms two oxides: SO2 and SO. • Sulfur forms a variety of compounds in which it has a +6, +4, +2, 0 or -2 oxidation state.
Group 17 (7A) - Halogens • This group consist of all nonmetals. • Form hydrogen halides (HX) that behave as strong acids in water, except for hydrogen fluoride. • Oxyacids of the halogens become stronger as the number of oxygen atoms attached to the halogen increase. • Interhalogens are compounds of two different halogens • Halogen-carbon compounds are important industrially: examples are Teflon, PVC and the Freons.
Group 18 (8A) – Noble Gases • Full valence shells ns2 np6 • Generally unreactive. • Krypton, xenon and radon will form compounds with the highly electronegative elements fluorine and oxygen.