Kaplan-Meier Estimator
Statistics2013 Poster. Kaplan-Meier Estimator. Bo Huang, Ching-Ray Yu and Christy Chuang-Stein Pfizer Inc. Statistics Saves Lives. The History of the Kaplan-Meier Estimator.

Kaplan-Meier Estimator
E N D
Presentation Transcript
Statistics2013 Poster Kaplan-Meier Estimator Bo Huang, Ching-Ray Yu and Christy Chuang-Stein Pfizer Inc. Statistics Saves Lives
The History of the Kaplan-Meier Estimator In a paper published in the Journal of the American Statistical Association in June 1958, Edward Kaplan and Paul Meier put forth a new, efficient method for estimating patient survival rates, taking into account the fact that some patients may have died during a research trial while others will survive beyond the end of the trial. The method, called the Kaplan-Meier estimator (also known as the product limit estimator), is based on a mathematical formula using information from those who have died and those who have survived to estimate the proportion of patients alive at any point during the trial. The estimator is plotted over time. The resulting curve is called the Kaplan-Meier curve, which is a series of horizontal steps of declining magnitude that, when a large enough sample is taken, approaches the true survival function for that population.
The Curve That Changed the World - John Easton • Paul Meier, 1924-2011, was the Ralph and Mary Otis Isham Distinguished Service Professor emeritus of statistics, pharmacological and physiological sciences, medicine. The 1958 paper has been cited more often, by a wide margin, than any other paper in the field of statistics. At last count it was the fifth most cited research paper of all time, in any field. It has been cited by another scientific publication about once, on average, for every day of Meier’s long life time. • The Kaplan-Meier estimator is used ubiquitously in medical studies to estimate and depict the fraction of patients who are alive at, for example one year, after the onset of a treatment. Estimating the survival curve is complicated by the fact that while some patients may still be alive at the end of a study, others may have dropped out of the study early. The latter are known as “censored observations” and are typically noted by tick marks on the estimated K-M curve. The Kaplan-Meier approach estimates the survival curve in the presence of censored observations • The concept of K-M estimator is equally applicable to other clinical endpoints such as the occurrence of a composite endpoint of vascular death, non-fatal MI and non-fatal stroke.
Edward L. Kaplan and Paul Meier According to Current Contents (1983), the seminal paper by Kaplan and Meier began in 1952 when Paul Meier, then at Johns Hopkins University, encountered Greenwood’s paper on the duration of cancer. A year later, while at Bell Laboratories, Kaplan became interested in the lifetimes of vacuum tubes in the repeaters in telephone cables buried in the ocean. Kaplan and Meier worked independently and submitted their respective work to the Journal of American Statistical Association. The journal encouraged them to submit a joint paper. Kaplan and Meier spent the next 4 years to resolve differences in their approaches and published a method that ultimately became the standard nonparametric approach for analyzing time to event data with censored observations.
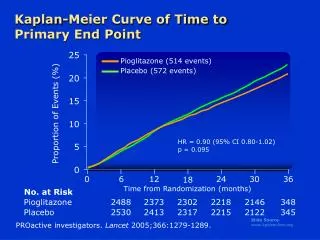
Example 1: KM curves of overall survival of an experimental drug vs the standard chemotherapy in a randomized phase III study in patients with advanced melanoma experimental drug standard chemotherapy Source: Marshall, Ribas and Huang, ASCO, 2010
Example 2: KM curve of progression-free survival in a randomized placebo-controlled phase III trial of sunitinib in patients with advanced pancreatic neuroendocrine tumors Source: Raymond et al., NEJM, 2011
Publications and Citations Since 1975 Publications refer to research papers related to the Kaplan-Meier method. Citations refer to all the medical and statistical papers that cite the 1958 seminal paper Publications and citations are on different scales in the double-axis figure Source: Microsoft academic research
Selected Quotes in Honor of Meier’s Work at His Death in 2011 The KM curve has become the standard tool used by medical researchers for determining the duration of survival in thousands of studies, ranging from cancer to AIDS to cardiovascular disease to diabetes, to name just a few -- New York Times (2011) The KM estimate was a very, very important advance. It seems so elementary now -- Washington Post (2011) Paul Meier’s work and the KM analysis have been responsible for saving millions of lives --- The Significance Magazine (2011) The KM estimator is used ubiquitously in medical studies to estimate and depict the fraction of patients living for a certain amount of time after treatment. It has since been applied to data from clinical trials of therapies for every disease from cancer to cardiology to concussion -- Science Life (2011)
Reference • Kaplan EL, Meier P. Nonparametric estimation from incomplete observations. J. Amer. Statist. Assn.53:457–481, 1958. • Kaplan EL. This week’s citation classic – Kaplan EL & Meier P. Nonparametric estimation from incomplete observations (J. Amer. Statis. Assn 53: 47-481, 1958). Current Contents. 24: 14, 1983. • Marshall M, Ribas A and Huang B. Evaluation of baseline serum C-reactive protein (CRP) and benefit from tremelimumab compared to chemotherapy in first-line melanoma. J. of Clin. Oncology, 2010 ASCO Annual Meeting Proceedings. 28: No 15: 2609, 2010. • Raymond E, Dahan L, Raoul JL, Bang YJ, Borbath I, Lombard-Bohas L, Valle J, Metrakos P, Smith D, Vinik A, Chen JS, Horsch D, Hammel P, Wiedenmann B, Van Cutsem E, Patyna S, Lu DR, Blanckmeister C, Chao R and Ruszniewski P. Sunitinibmalate for the treatment of pancreatic neuroendocrine tumors. NEJM.364: 501-13, 2011.
Reference (cont’d) • Hevesi D. Paul Meier, Statistician Who Revolutionized Drug Trials. New York Times. August 12, 2011 • Brown D. Paul Meier, biostatistician and co-inventor of a famous graph. The Washington Post. August 10, 2011 • Champkin J. Paul Meier - Statistician who saved millions of lives. Significance. August, 2011 • Easton J. The Curve That Changed the World. Science Life. August, 2011