Introduction:
Results, Discussion, and Conclusion (cont):

Introduction:
E N D
Presentation Transcript
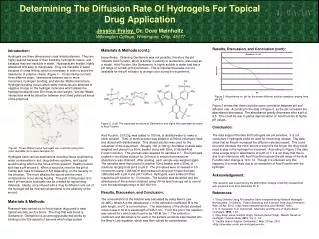
Results, Discussion, and Conclusion (cont): Figure 3 shows that there could be some correlation between pH and diffusion rate. According to the data in Figure 3, as the pH increased the absorbance decreased. The absorbance greatly decreases after a pH of 6.5. This could be due to partial deprotonation of Acid Fucshinat higher pH values. Conclusion: The data support the idea that hydrogels are pH sensitive. It is not conclusive if hydrogels could be used for timed drug release. The data show that as the pH increased the diffusion rate decreased. This could be useful because the more severe a wound is the longer the drug would need to stay in the hydrogel for treatment. According to Figure 3 the data show a largedrop in absorbance at a pH of 7; it is not likely that this is due to an interaction with Acid Fucshin because the pH range of the Acid Fucshincolor change is 12 to 14. Though it is unknown why this happens, it is most likely due to an interaction of Acid Fucshin with the hydrogel. Acknowledgement: This research was supported by the Wilmington College Chemistry Department with guidance from Dore Meinholtz Ph.D. References: 1. "Drug Delivery Using PH-sensitive Semi-interpenetrating Network Hydrogels - Northeastern University." Patent Searching and Invention Patenting Information. Web. 25 Apr. 2012. <http://www.freepatentsonline.com/5904927.html>. 2. K. S. Kazanskii, S. A. Dubrovskii. Chemistry and Physics of “Agricultural” Hydrogels. 1991, 1-37 3. Vijay Singh Jatav, Himmat Singh, Santosh Kumar Singh. Recent Trends on Hydrogel in Human Body.2011, Vol. 2, 1-6. 4. "Seattle Grace Hospital: Gentamicin." Web. 25 Apr. 2012. <http://sitemaker.umich.edu/mc5/gentamicin>. Materials & Methods (cont.): biosynthesis. Obtaining Gentamicin was not possible; therefore the pH indicator Acid Fucshin, which is similar in polarity to Gentamicin, was used as a model. Acid Fucshin, like Gentamicin, is highly soluble in water and has a pH range of 12(red)-pH14(colorless). This is important because it is not desirable for the pH indicator to change color during the experiment. Acid Fucshin, 0.0123g, was added to 1000mL of distilled water to make a stock solution. 75mL of stock solution was added to a 500mL volumetric flask and diluted with distilled water. This diluted solution was used for the remainder of the experiment. Roughly 100 (0.1901g) Soil Moist crystals were weighed and placed in a 50mL beaker along with 25mL of diluted Acid Fucshin solution. This process was repeated for samples 2-7. The hydrogels soaked in the diluted solution for 24 hours to ensure that maximum absorbance was obtained. After soaking, each sample was weighed again. The samples were then placed in another 50mL beaker with a 25mL of solutions ranging from pH 5 to pH 8. The pH values were increased in 0.5 increments using 1.0M NaOH and measured using a pH meter that was calibrated with a pH 4 and pH 7 buffers. Hydrogels, were soaked in their respective pH solution for 10 minutes. The solution was decanted and the absorbance of the solution obtained using UV-vis spectroscopy set to record over the wavelength range of 400-600 nm. Results, Discussion, and Conclusion: The concentration of the solution was calculated by using Beer’s Law (A=εBC), where A is the absorbance, ε is the extinction coefficient, B is the path length, and C is concentration. The absorbance of the diluted standard solution was 0.35 at a wavelength of 545.3 nm. From this point the equation was solved for ε which was found to be 190 M-1cm-1. The extinction coefficient and absorbance for each of the sample solutions was inserted into the Beer’s Law equation, which was then solved for concentration. Introduction: Hydrogels are three dimensional cross linked polymers. They are highly desired because of their flexibility, hydrophilic nature, and because they are insoluble in water. Hydrogels are flexible, highly absorbent and easy to manipulate. They are insoluble in water because of cross linking, which is necessary in order to avoid the dissolution of polymer chains (Figure 1). Cross linking can form three different ways: interactions between two or more monomers, hydrogen bonding, and Van der Waals interactions. Hydrogen bonding occurs when water molecules are attracted to negative charge on the hydrogel molecules which allows the hydrogel to absorb over 500 times its own weight. Van der Waals interactions work by attraction between short lived polarized areas of the polymers. Hydrogels have various applications including tissue engineering, water conservation in soil, drug delivery systems, and topical would healing which is the focus of this research. Healthy human skin has a pH around 5. When a wound is infection the pH of human skin rises to between 6.5-8 depending on the severity of the infection. The more alkaline the wound site the more complications occur during healing. The goal of this project is to see if pH sensitive hydrogels can be created for topical drug deleverly. Ideally, once infused with a drug its diffusion rate out of the hydrogel will be inversely proportional to the alkalinity of the wound. Materials & Methods: Research was carried out to find a typical drug used to treat topical wounds, such a burns. The drug that was found is Gentamicin. Gentamicin is an aminoglycoside that works by binding to the 30s subunit of bacteria which stops protein Determining The Diffusion Rate Of Hydrogels For Topical Drug ApplicationJessica Fraley, Dr. Dore MeinholtzWilmington College, Wilmington, Ohio, 45177 Figure 3. Absorbance vs. pH for the seven different solution samples ranging from pH 5-8. Figure 2. (Left) The expanded structure of Gentamicin and (right) the expanded structure of Acid Fucshin. Figure1. Three different ways hydrogels can cross link going from most desirable (A) to least desirable (C).