Microbial Count
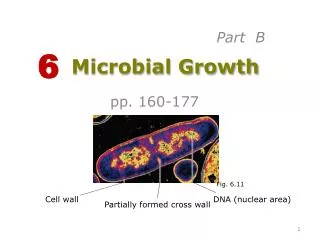
2) Counting chamber The most common method of enumerating the totalmicrobial cells is the direct counting of cell suspension ina counting chamber of known volume using amicroscope. There are many types of counting chambersfrom which the most common type is calledhemocytometer (originally d

Microbial Count
E N D
Presentation Transcript
1. Microbial Count Aim: Count the number of bacterial cells in a provided sample
Methods: Total count, Viable count
I. Total count
This technique involves the count of all bacteria in a
given suspension either viable or dead. This can be
performed either:
1) Turbidimetry
Total number of bacteria is calculated from a standard calibration curve which is obtained by plotting log bacterial count of a suspension against absorbance of this suspension.
2. 2) Counting chamber
The most common method of enumerating the total
microbial cells is the direct counting of cell suspension in
a counting chamber of known volume using a
microscope. There are many types of counting chambers
from which the most common type is called
hemocytometer (originally designed for performing
blood cell counts). It consists of a thick glass microscopic
slide with a rectangular groove that creates a chamber
of known dimensions.
3. This chamber is marked with a counting grid (network)
of perpendicular lines divided into:
- Nine large squares (1mm x 1mm)
- The center square is divided into 25 smaller squares
(0.2mm x 0.2mm).
- Depth of the counting chamber : 0.1 mm.
Therefore it is possible to count the number of cells in
a specific volume of fluid (10-20 ul in each slide notch),
and thereby calculate the concentration of cells in the
original fluid volume.
8. II. Viable count
Pour plate (seed) method
Principle
High numbers of bacteria are often present in samples.
Some samples may contain up to 1010 bacteria /ml. At
this concentration, a 0.1 ml volume plated onto a
nutrient agar plate could potentially yield one billion
colonies which is impossible to count in a Petri dish.
To overcome this problem, serial dilutions of the sample
prior to plating reduce the number of organisms such
that when a small volume of the diluted sample is plated,
the colonies are countable. For statistical accuracy, only
plates containing between 30 and 300 colonies are
counted.
9.
Procedure:
1. Make a serial dilution of the supplied bacterial culture by transferring aseptically exactly 1 ml amounts (using a sterile 1 ml pipette) into the sterile test tubes containing 9 ml saline solutions.
2. Each of the tubes must be thoroughly mixed (by means of a new 1 ml pipette) to insure even distribution of the organism.
3. With a sterile 1 ml pipette, aseptically transfer 1 ml of each dilution into each of 3 sterile Petri dishes. Mark each plate with the dilution present.
4. Melt the nutrient agar, pour into Petri dishes, and mix the bacterial suspension with the nutrient agar.
5. Incubate; count-the number of colonies choosing plates that have between 30 and 300 colonies.
11. Precautions for a succesful experiment:
1) Good mixing of the m.o. both in:
a) the 9-ml saline tubes
b) the Petri dishes together with melted nutrient agar.
2) Melted agar tubes must be poured at a temp. not too high (to avoid killing of the m.o.) & not too low (to allow for a sufficient mixing time that will insure an even distribution of the m.o. throughout the plate).
3) Petri dishes must be inverted upside-down immediately after solidification of poured nutrient agar to prevent condensed water vapor droplets in the inner side of the plate lid from falling-down over the agar surface, otherwise, wet agar surface will cause overlapping of the microbial colonies appearing as an uncountable patch over agar surface.
13. 2) Membrane filtration method
In this method cells are filtered onto membrane filter which is then placed over nutrient agar surface plates. Number of c.f.u/ml is determined after incubation at suitable temp. & time.