Parvalbumin as a Marker for Proprioceptive Axons in Developing Mouse Spinal Cord
This research highlights the use of parvalbumin as a reliable marker for proprioceptive axons during embryonic and early postnatal stages. Through dual immunolabeling with calbindin and VGLUT1, we demonstrated the distinct interactions of parvalbumin-immunoreactive axons with Renshaw cells and the absence of VGLUT1 in embryonic spinal cord. Our findings reveal a significant proliferation of axonal contacts postnatally and correlate well with anterograde tracing data. This approach provides clarity in studying the development of proprioceptive pathways.

Parvalbumin as a Marker for Proprioceptive Axons in Developing Mouse Spinal Cord
E N D
Presentation Transcript
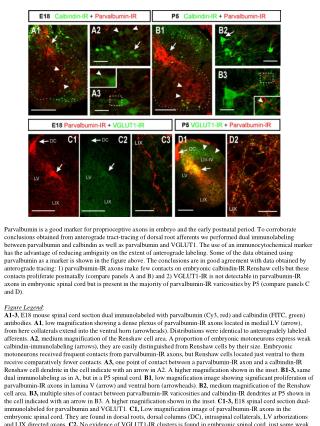
Parvalbumin is a good marker for proprioceptive axons in embryo and the early postnatal period. To corroborate conclusions obtained from anterograde tract-tracing of dorsal root afferents we performed dual immunolabeling between parvalbumin and calbindin as well as parvalbumin and VGLUT1. The use of an immunocytochemical marker has the advantage of reducing ambiguity on the extent of anterograde labeling. Some of the data obtained using parvalbumin as a marker is shown in the figure above. The conclusions are in good agreement with data obtained by anterograde tracing: 1) parvalbumin-IR axons make few contacts on embryonic calbindin-IR Renshaw cells but these contacts proliferate postnatally (compare panels A and B) and 2) VGLUT1-IR is not detectable in parvalbumin-IR axons in embryonic spinal cord but is present in the majority of parvalbumin-IR varicosities by P5 (compare panels C and D). Figure Legend: A1-3, E18 mouse spinal cord section dual immunolabeled with parvalbumin (Cy3, red) and calbindin (FITC, green) antibodies. A1, low magnification showing a dense plexus of parvalbumin-IR axons located in medial LV (arrow), from here collaterals extend into the ventral horn (arrowheads). Distributions were identical to anterogradely labeled afferents. A2, medium magnification of the Renshaw cell area. A proportion of embryonic motoneurons express weak calbindin-immunolabeling (arrows), they are easily distinguished from Renshaw cells by their size. Embryonic motoneurons received frequent contacts from parvalbumin-IR axons, but Renshaw cells located just ventral to them receive comparatively fewer contacts. A3, one point of contact between a parvalbumin-IR axon and a calbindin-IR Renshaw cell dendrite in the cell indicate with an arrow in A2. A higher magnification shown in the inset. B1-3, same dual immunolabeling as in A, but in a P5 spinal cord. B1, low magnification image showing significant proliferation of parvalbumin-IR axons in lamina V (arrow) and ventral horn (arrowheads). B2, medium magnification of the Renshaw cell area. B3, multiple sites of contact between parvalbumin-IR varicosities and calbindin-IR dendrites at P5 shown in the cell indicated with an arrow in B3. A higher magnification shown in the inset. C1-3, E18 spinal cord section dual-immunolabeled for parvalbumin and VGLUT1. C1, Low magnification image of parvalbumin-IR axons in the embryonic spinal cord. They are found in dorsal roots, dorsal columns (DC), intraspinal collaterals, LV arborizations and LIX directed axons. C2, No evidence of VGLUT1-IR clusters is found in embryonic spinal cord, just some weak and diffuse VGLUT1-immunoreactivity was noted in the LV region. C3, High magnification of a LIX parvalbumin-IR collateral with no evidence of VGLUT1-IR. D1, Parvalbumin and VGLUT1 dual immunolabeled section in a P5 spinal cord section at low magnification. A high density of intensely immunolabeled VGLUT1-IR varicosities is present in the LIII-LIV region and in the trajectory of proprioceptive axons in LV and LIX. D2, High magnification image of LIX showing VGLUT1-IR clusters inside the varicosities of parvalbumin-IR axons. Scale bars, in A1,B1,C1,C2,D1 200 µm; in A2,B2, 50 µm; in B, A3, B3, C3, D2, 20 µm.