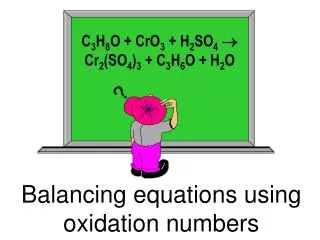
18.3 Balancing equations using Half-reactions in acidic or basic environments
90 likes | 277 Views
18.3 Balancing equations using Half-reactions in acidic or basic environments. Read pages 730-750. METHOD 1 – USING THE TABLE OF STANDARD REDUCTION POTENTIALS. identify reactants and products for possible half reactions. Find half reaction equations from page 11 (data booklet) that match

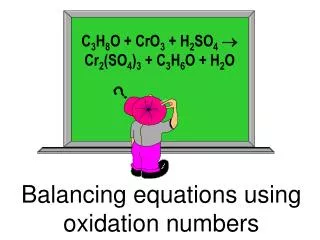
18.3 Balancing equations using Half-reactions in acidic or basic environments
E N D
Presentation Transcript
18.3 Balancing equations using Half-reactions in acidic or basic environments Read pages 730-750
METHOD 1 – USING THE TABLE OF STANDARDREDUCTION POTENTIALS • identify reactants and products for possible half reactions. • Find half reaction equations from page 11 (data booklet) that match • Look for a reduction half reaction and an oxidation half reaction. • Make the number of electrons lost equal the number of electrons gained. (multiply the reactions) • Add the oxidation and reduction half reactions. Electrons will cancel out and equation is balanced NOTE: • number of atoms and total charge on both sides must balance out. • If it takes place in acidic environment add H+, if basic environment add OH- to help balance out charges and atoms
SAMPLE PROBLEM 1 • IO3-(aq)+ Al (s) → I2 (s) + Al 3+(aq) Find half reactions: IO3-(aq) → I2 (s) Al (s) → Al 3+(aq)
try to find a reduction and an oxidation half reaction that matches on page 11 Reduction: 2IO3-(aq)+ 12H+ (aq) + 10 e-→ I2 (s) + 6H2O (aq) Oxidation: Al (s) → Al 3+(aq) + 3 e-
Multiply by 3 6IO3-(aq)+ 36H+ (aq) + 30 e-→ 3I2 (s) + 18H2O (aq) Multiply by 10 10Al (s) → 10Al 3+(aq) + 30 e- Finally, add them together and inspect. 6IO3-(aq)+ 36H+ (aq) + 10Al (s) → 3I2 (s) + 18H2O (aq) +10Al 3+(aq)
SAMPLE PROBLEM 2 • Use half reactions found on page 11to write a balanced redox equation for the reduction of acidic MnO4-(aq) solution to Mn 2+(aq) by Ni (s).
SAMPLE PROBLEM 3 • Use the half reactions found on page 11to write a balanced redox equation for the oxidation of NO (g) by an acidic Cr2O72-(aq) solution.
PRACTICE: Write the balanced oxidation-reduction equation for: • the reduction of acidic MnO4- (aq) solution to Mn 2+ (aq) by Fe 2+ (aq). • the reduction of acidic MnO4- (aq) solution to MnO2(s) by Pb (s). • Al + Fe 3+ → Al 3+ + Fe 2+ • the oxidation of Cl- (aq) by acidic H2O2(aq)
Homework • Page732 # 17-20 • Page 734 # 21-24