Chapter 7: Periodic Properties of the Elements
590 likes | 2.04k Views
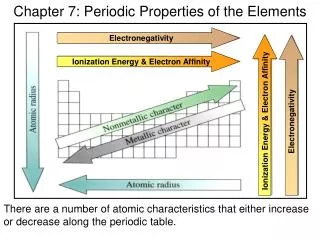
Electronegativity. Ionization Energy & Electron Affinity. Ionization Energy & Electron Affinity. Electronegativity. Chapter 7: Periodic Properties of the Elements. There are a number of atomic characteristics that either increase or decrease along the periodic table. Atomic Radius:

Chapter 7: Periodic Properties of the Elements
E N D
Presentation Transcript
Electronegativity Ionization Energy & Electron Affinity Ionization Energy & Electron Affinity Electronegativity Chapter 7: Periodic Properties of the Elements There are a number of atomic characteristics that either increase or decrease along the periodic table.
Atomic Radius: As you go down a group, the atomic radius increases with increasing energy level. Atomic radius decreases as you go left to right along a period because the greater nuclear charge pulls the electrons in closer to the nucleus. s-block p-block
- + Why ionization energy decreases and atomic/ionic radius increases as go down a group: Shielding effect: The inner electron shells insulate the valence electrons from some of the electrical attraction with the positive charge of the nucleus. valence e- nucleus core electrons
! ! - - - - - + + + + + + + Why ionization energy increases and atomic/ionic radius decreases as go across a period: Increasing Effective Nuclear charge(Zeff): Electrons in the outermost energy levels do not effectively screen each other from an increasingly positive nucleus. nucleus Zeff = Z - S Li: Zeff = 3 – 2 = 1 N: Zeff = 7 – 2 = 5
A few definitions: van der Waals radius: The nonbonding radius of atoms Covalent radius: The radius of atoms covalently bonded to another atom; is smaller than the van der Waals radius Isoelectronic: Different ions that have the same number of electrons Ex: O2-, F-, Na+, Mg2+, Al3+ all have 10 electrons Ionization energy: The amount of energy required to remove an electron from an atom or ion. Electron affinity: The amount of energy released when an electron is added to an atom or ion.
There is a spike in ionization energy whenever a noble gas electron configuration is disrupted. Q: What is the valence electron configuration for an atom that has the following ionization energies: 1st: 734 kJ/mol, 2nd: 1850 kJ/mol, 3rd: 16,432 kJ/mol A: ns2 Large spike in IE indicates noble gas core is disrupted
Electron Affinity:the ability of an atom to gain an electron. This is closely related to ionization energy, and increases going left to right, and decreases going down.
Electronegativity: Ability of an atom to attract electrons when in a molecule.
Electronegativity increases going left to right, and decreases going down.
Fe3+: 4s 3d Electron configurations of ions • If electrons are added to make an anion, they fill the lowest energy levels first. (Auf bau principle) • If electrons are removed to make a cation, the are taken from the highest energy levels first. Atom Ion 1s2 2s1 1s2 2s0 Li+ Li Fe [Ar] 4s2 3d6 [Ar] 4s0 3d6 Fe2+ Notice that the Fe3+ ion has the maximum multiplicity possible. Hence, its greater stability wrt Fe2+ Fe3+ [Ar] 4s0 3d5
Electron configuration exceptions and their ions Atom Ion [Ar] 4s1 3d5 [Ar] 4s0 3d5 Cr+ Cr Cr3+ [Ar] 4s0 3d3 [Ar] 4s0 3d0 Cr6+ [Ar] 4s1 3d10 [Ar] 4s0 3d10 Cu+ Cu [Ar] 4s0 3d9 Cu2+ Au [Xe] 6s1 4f14 5d10 [Xe] 6s0 4f14 5d10 Au+ Au3+ [Xe] 6s0 4f14 5d8
Characteristic Properties of Metals and Nonmetals Metals Metalloids Nonmetals • Have a shiny luster and are usually silvery in color • Do not have luster; various colors Au Cu Notable exceptions: • Solids are usually brittle • Solids are malleable and ductile • Poor conductors of heat and electricity Hg is only liquid metal at RT • Most nonmetal oxides are molecular solids that form acidic solutions. • Good conductors of heat and electricity • Metal oxides are ionic solids and form basic solutions • Tend to form anions or oxyanions in solution • Form cations in solution
P S Cu Au I F Pale yellow gas Light green gas Dark orange liquid/gas Dark violet crystals/gas Elements with Color red Metals Nonmetals white Halogens
Properties of SOLID ionic compounds (M + NM) • Poor conductors of electricity and heat. • Generally high melting (more than 150°C). • Crystalline, hard and brittle. NaCl crystal lattice structure
Properties of ionic compounds (cont.) • Molten ionic compounds form liquids that are an electrical conductors. • Ionic solids that are water soluble, dissolve to form solutions that are electrical conductors. • The solubility of ionic compounds depends upon the lattice energy. The greater the lattice energy, the lower the solubility. - +
Acid-Base Behavior of Oxides • Most metal oxides form basic solutions • Most nonmetal oxides form acidic solutions • The acidity of the solution increases with oxidation number of the central atom pH: SO3(aq) SO2(aq) < Hint: an easy way to evaluate the acidity is that pH as the # oxygen atoms • Amphoteric substances can act as either an acid or a base
Group Trends Alkali metals: low IE highly reactive, soft silvery metals with a low density and low melting point. While lithium reacts with oxygen to form lithium oxide 4 Li + O2 2 Li2O the other alkali metals form peroxides. (peroxide = O22-) 2 Na + O2 Na2O2 Potassium, rubidium and cesium react with oxygen to form superoxides (superoxide = O2) K + O2 KO2
Alkaline earth metals • Harder, more dense and have a higher melting point than Group 1 metals. • 1st IE is low, but not as low as Group 1 because disrupting pseudo-noble gas configuration (s2) • Increasing reactivity with increasing atomic number due to increase in nuclear shielding. • Beryllium will not react with water, but the other alkaline earth metals will to form the metal hydroxide and hydrogen gas. Ca + 2 H2O Ca(OH)2 + H2