Packing, Cavities
Packing, Cavities. atomic radii, contact-distance profiles cavities P-P interactions crystal contacts solvent channels de-stabilizing mutations in core (TS mutants?) entropy effects on surface. Surface Calculations. Lee & Richards - solvent accessible surface

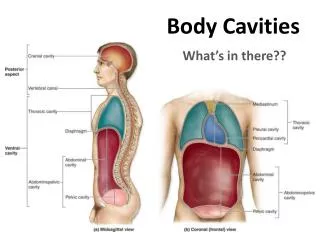
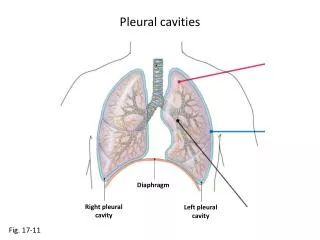
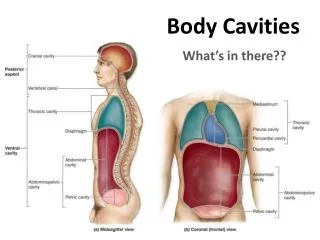
Packing, Cavities
E N D
Presentation Transcript
Packing, Cavities • atomic radii, contact-distance profiles • cavities • P-P interactions • crystal contacts • solvent channels • de-stabilizing mutations in core (TS mutants?) • entropy effects on surface
Surface Calculations • Lee & Richards - solvent accessible surface • expanded atom spheres, reentrant surfaces • typical water probe radius: 1.4A • computation: grid points vs. tangents (algebraic/analytic)
Alpha-shape theory • Voronoi methods • Liang and Edelsbrunner • pockets, pockets, depressions – depends on width of opening
Packing Density from Richards (1977) crambin (blue=vdw, red=interstitial) data on compressibility?
Jie Liang and Ken A. Dill (BiophysJ, 2001). Are Proteins Well-Packed? • 636 proteins; 1.4A probe radius • proteins are dense (like solids), yet atoms are arranged like liquids (without voids) • P=0.76 for hex-packed spheres • P=0.74 for protein interiors • distribution of number/size of voids is more variable, like a liquid • surface area scales linearly with volume, instead of A a V-2/3
Clefts/Active Sites • Liang Edelsbrunner, Woodward (1998)
De-stabilizing mutations in core • cavities, tolerance, re-packing • Serrano L., Kellis J., Cann P., Matouschek A. & Fersht A. (1992) • In barnase, 15 mutants were constructed in which a hydrophobic interaction was deleted • strong correlation between the degree of destabilization (which ranges from 0.60 to 4.71 kcal/mol) and the number of methylene groups deleted • average free energy decrease for removal of a completely buried methylene group was found to be 1.5±0.6 kcal/mol. This is additive. • double-mutants? • temperature-sensitive mutants?
Side-chain contact profiles • Sippl – knowledge-based potentials • Subramaniam – PDF’s • dependence: radial distance, sequence separation
Protein-protein interactions • flat and hydrophobic? • Janin • Jones and Thornton (1996), PNAS – data on flatness, H-bonds • which is predominant: H-bonds vs. salt-bridges vs. hydrophobic interactions? (homodimers)
Complementarity of P-P interfaces • shape complementarity • measure “gaps” or voids • cavities at interfaces (Hubbard and Argos, 1994) • more common than in core • suggests complementarity doesn’t have to be perfect • surface normals: • R Norel, SL Lin, HL Wolfson and R Nussinov
LoConte, Chothia, and Janin (1999), JMB. • The average interface has approximately the same non-polar character as the protein surface as a whole, and carries somewhat fewer charged groups. • However, some interfaces are significantly more polar and others more non-polar than the average. • 1/3 of interface atoms becomes completely buried; packing density is similar to core (like organic solids) • in high-res structures, remainder of space is filled in by water molecules (making H-bonds) • size for “typical” interfaces: 1600±400Å2
Electrostatic Complementarity • McCoy et al. (1997) • defined two correlation coefficients between surfaces (summed over contacts): charge complementarity (ion pairs), and electrostatic potentials • depends on assignment of partial charges, solvation... • examine effect on DG • charge correlations: -0.1..+0.1 (insignificant) • electrostatic potential correlations: 0.1..0.7 (significant) • steering and diffusion (Kozak et al., 1995)
Jones and Thornton – Patch Analysis, PP-interface prediction
Examples of P-P interactions • b-lactamase/BLIP – one of the tightest • antibody-antigens (HYHel5) • SH2/SH3 and tyrosine kinases • PDZ domains • calmodulin • proteases, kinases (recognize+catalyze)
beta-sheet extension • arylamine N-acetyltransferase (nat) • acetylates isoniazid in M.smegmatis • pdb: 1W6F • active in solution as both monomer and dimer • lower surface area, but many H-bonds
PPI Trivia • obligate vs. transient complexes - affinity • differences between antigen-antibody, protease-inhibitor, and rest of complexes • induced conformational changes • allostery • evolutionary conservation at interfaces (Caffrey et al. 2004), • correlated mutations? mutational hot spots, evolutionary trace (Lichtarge) • why are homodimers so common? (Lukatsky et al, 2007) succinyl-CoA synthetase green=contact purple=conserved
Crystal-lattice Contacts • Carugo and Argos (1997) • small: 45%<100Å2, 8%>500Å2 • properties like rest of surface, so probably random • induced changes (rms)?
Protein-Protein Docking • FTDOCK - Gabb, Jackson, and Sternberg (1997) • use Fourier transform to evaluate shape correlation function • correlation function includes shape and electrostatic complementarity of surfaces • try 6912 rotations, Da=15º
r=-15 grid nodes within 1.8A of protein atom are “inside” • Multidock (Jackson, Gabb, Sternberg, 1998) • what about induced fit? alternative side-chain rotamers? domain rotations? • need refinement to do scoring of complexes, increase sensitivity to recognize correct interaction • add term for solvation energy (soft-sphere Langevin interactions between solvent grid points and surface side-chains) • sample different rotamers • Betts & Sternberg (1999) – induced fit at interfaces side-chain and backbone movements
GRAMM (Vakser) • low resolution protein docking • maybe removing details will help... • search 6D space for maximal surface overlap (20º rotations) • intermolecular overlap function • evaluated on a coarse 7Å grid PatchDock, FireDock (Nussinov & Wolfson)