Isotopes and Mass Number
210 likes | 618 Views
Isotopes and Mass Number. Leon the Electron. Jimmy Neutron. Atomic Number. The number of protons in each atom identifies it as an atom of a particular element Each atom has a unique number of protons and, in effect, the number of protons cannot change for a particular atom

Isotopes and Mass Number
E N D
Presentation Transcript
Isotopes and Mass Number Leon the Electron Jimmy Neutron
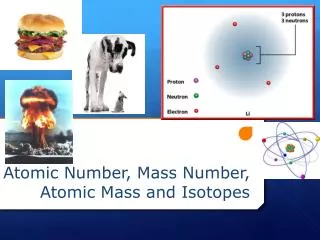
Atomic Number • The number of protons in each atom identifies it as an atom of a particular element • Each atom has a unique number of protons and, in effect, the number of protons cannot change for a particular atom • The Periodic Table is organized by increasing atomic number (left-to-right and top-to-bottom) • All atoms are neutral, so the # protons = the # of electrons because the number of positive charges equals the number of negative charges • Atomic # = # of protons = # electrons • For Helium, the atomic # is 2. So, weknow that Helium also has 2 protons and 2 electrons
Determining the State of Matter of a Particular Element Using the Periodic Table
How Atoms Differ The order of the elements on the Periodic Table is based on the number of protons in the nucleus. Protons are the only thing in an atom that does not change.
Proton, Neutron, Electron Practice How many protons does Platinum have? How many neutrons does Mercury have? How many electrons does Lead have? What similarities do these three elements share? The number of protons in the nucleus is called the atomic number. Also since the positive charges must equal the negative charges to have a neutral atom, and all atoms are neutral, the atomic number will also equal the number of electrons in an atom. What is the atomic number of this atom? What is the element?
Isotopes Don't be an isodope, learn about an isotope. All atoms of a particular element have the same number of protons and electrons The number of neutrons may differ In nature most elements are found as a mixture of isotopes
Isotopes and Mass • Isotopes differ in mass • Isotopes of an atom have the same chemical behavior. Why? ?
Mass Number • Identifies each of the isotopes of an element 10747Ag 10947Ag Mass number = top Atomic number = bottom # neutrons = mass # - atomic #
Banana Isotope • 93.25% of K atoms have 20 neutrons • 6.7302% of K atoms have 22 neutrons • .0117% of K atoms have 21 neutrons
Mass of Individual Atoms • The atomic mass of an element is the weighted average mass of the isotopes of that element • To determine the weighted average atomic mass of an element, calculate the mass contribution of each of the isotopes and add the values together
Weighted Average Atomic Mass 3517Cl 3717Cl Atomic mass: 34.969 amu Atomic mass: 36.966 amu Percent abundance:75.770% % abundance: 24.230% Mass Contribution: Mass Contribution: (34.969)(.75770)= 26.496 amu (36.966)(.24230)=8.957 amu Weighted average atomic mass of chlorine = (26.496 amu + 8.957 amu) = 35.453 amu
Steps to Solving Weighted Averages of Isotopes 1. Turn the % into a decimal for the % abundance. 2. Multiply the decimal by the atomic mass of each particular isotope. 3. Add the masses of the isotopes together. 4. Your answer will be in amu’s (atomic mass units).
You try one….. Calculate the atomic mass of Magnesium. The three Magnesium isotopes have atomic masses and relative abundances of 23.985 amu (79.99%), 24.986 amu (10.00%), and 25.982 amu (11.01%).
Weighted Averages of Isotopes Practice I 1. What is the atomic mass of this sample of Silver? 107 Ag 106.905 amu 52 % 109 Ag 108.905 amu 48 % 2. What is the average atomic mass of this sample of Hydrogen? 1H 12 % 2H 48 % 3H 40 %
Weighted Averages of Isotopes Practice I Continued 3. Boron has two naturally occurring isotopes, Boron-10 (abundance 19.8%, mass = 10.013 amu, Boron –11 ( abundance 80.2%, mass = 11.009 amu). Calculate the average atomic mass of Boron.