Maximizing Heat Exchange Capacity in Thermochemistry: Understanding Changes and State Functions
Explore the fundamental concepts of thermochemistry focusing on changes in total heat capacity, internal energy, Enthalpy, State Functions, and Heat of Formation. Learn about the significance of heat exchange, enthalpy change, and application of Hess’s Law in heat reactions. Discover how to calculate heat exchange between a system and surroundings, processes of forming substances from elements, and state function equations for a better understanding of thermodynamic changes.

Maximizing Heat Exchange Capacity in Thermochemistry: Understanding Changes and State Functions
E N D
Presentation Transcript
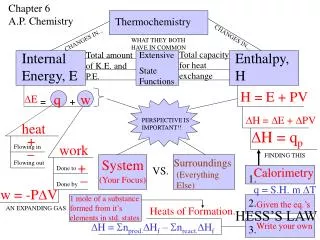
Chapter 6 A.P. Chemistry Thermochemistry CHANGES IN... WHAT THEY BOTH HAVE IN COMMON CHANGES IN... Total capacity for heat exchange Internal Energy, E Total amount of K.E. and P.E. Extensive Enthalpy, H State Functions H = E + PV q w DE = + DH = DE + DPV PERSPECTIVE IS IMPORTANT!! heat DH = qp + _ work Flowing in FINDING THIS System Surroundings Flowing out + Done to VS. Calorimetry _ (Everything Else) 1. (Your Focus) Done by q = S.H. m DT w = -PDV 1 mole of a substance formed from it’s elements in std. states 2. Given the eq.’s AN EXPANDING GAS Heats of Formation. HESS’S LAW DH = Snprod.DHf – Snreact.DHf Write your own 3.