Understanding Resonance in Chemical Structures: A Detailed Guide
80 likes | 202 Views
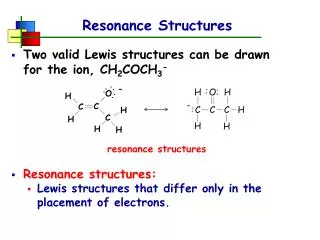
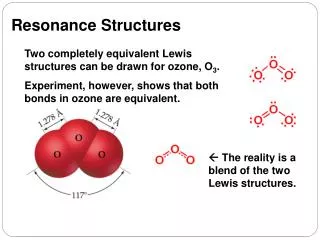
This guide explores the concept of resonance in chemical structures, focusing on octet rule compliance for various molecules such as [HCO2]⁻, [SO3]²⁻, and N2O. It outlines steps for skeletal structure drawing, electron counting, and formal charge minimization. Readers will learn how to identify central atoms, add electron pairs to achieve octets, introduce double bonds when necessary, and draw resonance structures that represent equally likely configurations. The guide emphasizes best practices for constructing and interpreting line structures, ensuring clarity in chemical representations.

Understanding Resonance in Chemical Structures: A Detailed Guide
E N D
Presentation Transcript
[HCO2]- • Any common octet breaking atoms? • How many electrons are present? • What is the central atom? • Draw a skeletal structure • Add electrons: do you have enough to give all atoms octets? • Yes? Then minimize formal charges if needed. • No? Then add double bonds until you have enough electrons. • Is it one that is typically found to have less? • Is there any reason you chose those double bonds rather than placing it elsewhere? • No? Then draw resonance structures to show all equally likely structures • Label each with the formal charge
O3 • Any common octet breaking atoms? • How many electrons are present? • What is the central atom? • Draw a skeletal structure • Add electrons: do you have enough to give all atoms octets? • Yes? Then minimize formal charges if needed. • No? Then add double bonds until you have enough electrons. • Is it one that is typically found to have less? • Is there any reason you chose those double bonds rather than placing it elsewhere? • No? Then draw resonance structures to show all equally likely structures • Label each with the formal charge
[SO3]2- • Any common octet breaking atoms? • How many electrons are present? • What is the central atom? • Draw a skeletal structure • Add electrons: do you have enough to give all atoms octets? • Yes? Then minimize formal charges if needed. • No? Then add double bonds until you have enough electrons. • Is it one that is typically found to have less? • Is there any reason you chose those double bonds rather than placing it elsewhere? • No? Then draw resonance structures to show all equally likely structures • Label each with the formal charge
Back through N2O • Any common octet breaking atoms? • How many electrons are present? • What is the central atom? • Draw a skeletal structure • Add electrons: do you have enough to give all atoms octets? • Yes? Then minimize formal charges if needed. • No? Then add double bonds until you have enough electrons. • Is it one that is typically found to have less? • Is there any reason you chose those double bonds rather than placing it elsewhere? • No? Then draw resonance structures to show all equally likely structures • Label each with the formal charge
Line Structures: CH3CHCH2COOH • Rules • The end of a line, or a corner is a carbon • All hydrogens attached to a carbon are assumed to be there. Do not draw them. • Don’t forget the three hydrogens attached to the carbon at an end of a line are there! • Heteroatoms: aka atoms other than C and H, need to be written in. • All hydrogens attached to heteroatoms must be drawn in.
Line Structures: NH2CH2CH(OH)CH3 • Rules • The end of a line, or a corner is a carbon • All hydrogens attached to a carbon are assumed to be there. Do not draw them. • Don’t forget the three hydrogens attached to the carbon at an end of a line are there! • Heteroatoms: aka atoms other than C and H, need to be written in. • All hydrogens attached to heteroatoms must be drawn in.
Benzene • C6H6 • 6-membered ring • Draw the lewis structure • Be sure to include any resonance structures • Draw the line structure • Be sure to include any resonance structures