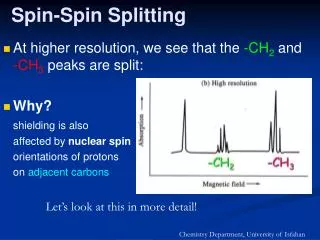
1 st -order spin-spin coupling
1 st -order spin-spin coupling. We observe 1 st -order NMR spectra when the frequency difference between the chemical shifts of any given pair of nuclei is much larger than the value of the coupling constant between them Dn / J > 10

1 st -order spin-spin coupling
E N D
Presentation Transcript
1st-order spin-spin coupling We observe 1st-order NMR spectra when the frequency difference between the chemical shifts of any given pair of nuclei is much larger than the value of the coupling constant between them Dn/J > 10 and any set of chemically equivalent nuclei is also magnetically equivalent. • 1st-order NMR spectra exhibit a number of simple characteristics: • Multiplicities that result from coupling reflect the 2nI + 1 rule (IH = ½). • The intensities of spin-spin multiplets correspond to Pascal’s triangle for I = 1/2 • Nuclei with the same chemical shift do not split each other, even when the coupling constant between them is not zero. • Spacings between adjacent components of a spin-spin multiplet are equal to J. • Spin-spin multiplets are centred on the resonance frequency
2nd-order spin-spin coupling We observe 2nd-order NMR spectra when the frequency difference between the chemical shifts of any given pair of nuclei is small compared to the value of the coupling constant between them Dn/J < 10 and/or any set of chemically equivalent nuclei is not magnetically equivalent. Nuclei are chemically equivalent if they can be interchanged by a symmetry operation of the molecule. Nuclei that are interchangeable by a rotation (Cn) are said to be homotopic. Nuclei related only by a mirror plane are termed enantiotopic. Chemically equivalent nuclei are isochronous (same chemical shift) but the converse is not necessarily true. Nuclei are magnetically equivalent if they are isochronous and if all the coupling constants for couplings to any other nucleus are equal for each nucleus (isogamous coupling).
Enantiotopic and Diastereotopic Protons Enantiotopic protons by inversion (i) Diastereotopic protons of methylene groups plane makes H1’s and H2’s equivalent no plane through CH2’s thus the protons are diastereotopic chiral molecule achiral molecule Diastereotopic protons can not be placed in same chemical environment
Staggered Rotamers – non-chiral gauche anti anti rotamer: H1 and H2 as well as H3 and H4 are enantiotopic interchanged through a plane of symmetry other rotamers: no symmetry, H1 and H2 as well as H3 and H4 arediastereotopic get chemical shifts for the anti and gauche rotamers rapid rotation gives one chemical shift for H1 and H2 and another for H3 and H4
Staggered Rotamers – chiral centre next to methylene group • H1 and H2 are not equivalent as cannot be interchanged by a symmetry operation • no plane, axis or inversion center • not interchanged by rapid rotation • averaged chemical shift is not identical
Magnetic Equivalence If chemical shift equivalent nuclei couple equally to other nuclei then they are magnetically equivalent magnetic equivalent if symmetrically disposed with respect to each nuclei in the spin system. H1 and H1’ chemical shift equivalent J12 J12’ = 7-10 Hz, J1’2 J1’2’ = 1 Hz since H1 and H1’ couple differently to H2magnetically not equivalent AA’XX’ spectrum is complex
Spectrum of 1-Chloro-4-nitrobenzene AA’XX’ spectrum
AB spin systems DnAB DnAB DnAB DnAB DnAB DnAB DnAB DnAB DnAB
Analysis of AB spin systems DnAB = (4C2 – J2) The ratio of intensities between larger inner and smaller outer peaks is (1+J/2C)/(1-J/2C)
Geminal couplings Geminal (2J) couplings are usually negative and usually reach values of up to 30 Hz. Geminal protons attached to double and triple bonds can have positive coupling constants. Lone pairs of electrons can donate electron density and make 2J more positive. Coupling of magnetically equivalent protons do not appear in the NMR spectrum but the coupling constants can be determined by deuteriation or from 13C satellites.
Vicinal Couplings Vicinal (3J) couplings are often positive and usually reach values of up to 20 Hz.
Long-Range Couplings All couplings between protons that are more than 3 bonds apart are called long-range couplings (4J, 5J, etc.). Their coupling constant can reach values between 0.5-3 Hz if both sets of protons are connected to the same p-electron system.