10.3 Strong and Weak Bases
40 likes | 165 Views
Learn the distinction between strong and weak bases, their ionization in water, and how they react differently. Compare their properties like pH, conductivity, and reaction rates.

10.3 Strong and Weak Bases
E N D
Presentation Transcript
10.3 Strong and Weak Bases LO: I understand the difference between strong and weak bases.
10.3.1 Strong Bases A base is a substance that an acid. neutralises A strong base is a substance that is fully ionised to give ions when dissolved in water. hydroxide e.g. sodium hydroxide: NaOH(s) + water Na+(aq) + OH-(aq) LO: I understand the difference between strong and weak bases.
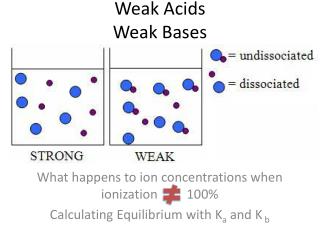
10.3.2 Weak Bases A weak base is a substance that is only ionised to give hydroxide ions when dissolved in water. partially e.g. ammonia solution: NH3(aq) + H2O(l) NH4+(aq) + OH-(aq) 99.6% of the ammonia remains as . covalent molecules The equilibrium lies very much to the . left LO: I understand the difference between strong and weak bases.
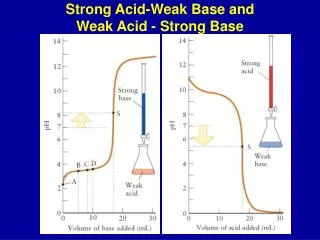
10.3.3 Comparing Strong and Weak Bases Reactions of Bases In comparison to a strong base, a weak base: • Has a pH lower lower • Has a conductivity slower • Reacts at a rate. The stoichiometry is for equimolar strong and weak bases. identical LO: I understand the difference between strong and weak bases.