Understanding Properties of Matter: Atoms, Elements, and Bunsen Burners
1.33k likes | 1.49k Views
In this unit, students explore the fundamental concepts of matter, including atoms, elements, and the differences between pure substances and mixtures. Activities include discussing molecular behavior during phase changes, learning to use Bunsen burners safely, and completing various tasks that reinforce vocabulary and key ideas in physical science. The week's agenda includes quizzes, hands-on practice with Bunsen burners, and reflection on notable themes in Newton's life, enhancing students’ understanding of scientific principles and lab safety.

Understanding Properties of Matter: Atoms, Elements, and Bunsen Burners
E N D
Presentation Transcript
Physical Science CHS 2013-14 Unit 3 Properties of Matter
12/2/13Week of 12/2 to 12/6 • Entry Task • Find a definition for ATOMS and ELEMENT in the textbook.
Today’s Agenda • Get out your Newton Movie Questions. • Read through them together so that you know what you are listening for. • Raise your hand to share if you hear an answer to one of the questions. • The movie will be paused briefly when answers are heard.
12/2/13 • Exit Task • Describe some themes in Newton’s life.
12/3/13 • Entry Task • What is a pure substance? • What is a mixture? • List physical means of separation (p. 181). • 5 minute review and Turn in whatever you have for the Newton Questions.
C-notes on pages 178-182 and 190-194 Vocabulary (left) – meanings (right) Key Ideas (left) – answer (right) Describe the differences between pure substances and mixtures (a venn diagram may help). List the 4 phases of matter in order of increasing temperature Describe what happens at the molecular level when a substance melts. Describe what happens at the molecular level when a substance boils. • Element • Compound • Atom • Molecule • Mixture • Pure Substance • Solid • Liquid • Gas • Plasma • Intermolecular Forces • Melting Point • Boiling Point
C-notes on pages 178-182 and 190-194 Vocabulary (left) – meanings (right) Key Ideas (left) – answer (right) Describe the differences between pure substances and mixtures (a venn diagram may help). List the 4 phases of matter in order of increasing temperature Describe what happens at the molecular level when a substance melts. Describe what happens at the molecular level when a substance boils. • Element • Compound • Atom • Molecule • Mixture • Pure Substance • Solid • Liquid • Gas • Plasma • Intermolecular Forces • Melting Point • Boiling Point
12/3/13 • Exit Task • Explain the difference between an element and a compound.
12/4/13 • Entry Task • Look at your safety rules (Unit 1). • Write down 3 safety rules for using bunsen burners or open flames in the lab. • TEST MAKE-UPS: If you still need to complete a Unit 2 test YOU ARE RESPONSIBLE to schedule with me.
Today’s Agenda – Learn about Bunsen Burners • What is a Bunsen Burner - history • Video Tutorial – the one minute version • Slow down! – How to Light a Bunsen Burner • Notes – parts and steps of use • Tomorrow you will take a quiz and a performance assessment on using Bunsen burners. • You will have to pass the quiz and the performance assessment in order to get your bunsen burner license. • What is the right sequence?
Label the parts • air intake • combustion nozzle • barrel/body tube • gas jet (point to inside) • base • gas valve/dial
Functions of Parts • air intake: adjustable; controls amount of oxygen; adding more air makes flame hotter • combustion nozzle: gas and air mix to ignite • barrel: turn this to adjust air intake • gas valve/dial: adjustable; controls amount of gas entering burner; adding more gas makes flame taller • base: holds up Bunsen burner • gas jet: small hole that gas goes through
Steps to Light and Turn Off • Wear safety goggles, tie back long hair, no loose clothing. • Check striker for sparks. • Attach hose to burner and gas line . • Turn barrel clockwise to close and then open slightly to let a small amount of air flow. • Turn gas valve/dial counter clockwise to close. • turn on gas by rotating valve parallel to hose. • Open gas valve/dial slightly – should hear faint hissing. • Hold striker at 45 degree angle and light. • For a taller flame – open gas valve/dial more to allow more gas. For a shorter flame – turn gas valve/dial back counter clockwise. • For a hotter flame – turn barrel counter clockwise (increases air flow). Flame should turn blue. • For a cooler flame – turn barrel clockwise (decreases air flow). Flame should turn orange. • The ideal flame is pale blue with a darker blue core. Draw a picture of the flame. • To turn off: turn barrel clockwise for a cooler, orange flame. Completely turn off the gas valve/dial. Turn off gas line.
Safety Precautions • Wear safety goggles, tie back long hair, no loose clothing. • Hold Bunsen burner at the bottom to avoid burns. • Never leave open flames unattended. • Know location of safety exits, emergency shut-off and fire extinguishers.
12/4/13 • Exit Task • What part of the Bunsen Burner flame is the hottest? Draw a picture or explain
12/5/13 • Entry Task • Fill in the blanks: • Attach hose to _______ and ________ . • Turn barrel _________ to close and then open slightly to let a small amount of air flow. • Turn __________ _______-_______ to close. • turn on gas by rotating valve _________ to hose. • Open gas valve/dial slightly – should hear __________. • Hold _________ at 45 degree angle and light.
Today’s Agenda • TURN IN HOMEWORK ASSIGNMENT. • Complete notes • Take written portion of quiz. • Passed the quiz? • Practice the steps to light the burner and turn off the burner AT YOUR TABLE WITHOUT ACTUALLY USING GAS • Performance Assessment of these steps tomorrow or Monday.
12/5/13 • Exit Task-SKIPPED DUE TO QUIZ • Fill in the blanks: • To turn off: turn barrel _________ for a cooler, orange flame. Completely turn off the __________. Turn off _________.
12/6/13 • Entry Task • Say Something Nice
Today’s Agenda • Unit2 MC tests will be returned to you. • Please get out your red sheets. • Graph your post-assessment score. • Did you meet your goal set towards the beginning of the unit? • Unit 2 Math tests will be returned to you. • REQUIRED REFLECTIONS: If your Scantron score is less than 28. • Discuss Answers.
12/6/13 • Exit Task • Trade and Grade • Please turn in your Entry/Exit task sheets for this week: 12/2 – 12/6/13.
12/9/13Week of 12/9 to 12/13 • Entry Task • Papers handed back. • Add together your Unit 2 MC test POINTS and math portion POINTS. • Take the TOTAL POINTS and divide by 66. Multiply by 100 to calculate your percent. • Record this percent and write down if you would like to regain some points with a reflection.
Today’s Agenda • Review Math answers to Unit 2 test. • Divide into 3 groups: • Group 1 - reflecting on MC portion of UNIT 2 – retrieve your test from counter; sit down with separate sheet of paper for reflection. TESTS WILL BE RECOLLECTED AT END OF CLASS. • Group 2 – reflecting on MATH portion of UNIT 2. Remember to reflect on a separate piece of paper. • Group 3 – no reflection necessary. Read pages 200-210. Answer questions #1-10 on p. 211.
12/9/13 • Exit Task • Make a connection: Consider the amount of homework you completed for Unit 2. Also consider the quality of your efforts studying. Did your effort yield results in your test scores? Explain what these results show you.
12/10/13 • Entry Task • What kind of energy is heat? Does heat flow from hot to cold or cold to hot? • Homework: Read pages 200-210. Answer questions #1-10 on p. 211. Due Thursday 12/12/13 • SAFETY LECTURE!!
Today’s Agenda • On back of quiz: Please explain WHY the steps are in the SAFEST ORDER. • REVISE ANY CIRCLED items on the written Bunsen Burner. You must get 100% • PRACTICE: • 1. Goggles and tie back hair. • 2. attach hoses • 3. allow a small amount of air • 4. Close gas valve/dial • 5. Turn on gas line. • 6. Turn on small amount of gas. • 7. Light with striker • Performance Assessment for Bunsen Burners • Once you receive your license…. • Flame Test Lab – using colors to determine chemical elements. NEVER LEAVE FLAME UNATTENDED.
Today’s Agenda • On back of quiz: Please explain WHY the steps are in the SAFEST ORDER. • REVISE ANY CIRCLED items on the written Bunsen Burner. You must get 100% • PRACTICE: • 1. Goggles and tie back hair. • 2. attach hoses • 3. allow a small amount of air • 4. Close gas valve/dial • 5. Turn on gas line. • 6. Turn on small amount of gas. • 7. Light with striker • Performance Assessment for Bunsen Burners • Once you receive your license…. • Flame Test Lab – using colors to determine chemical elements. NEVER LEAVE FLAME UNATTENDED.
12/10/13 • Exit Task • CANCELLED DUE TO PERFORMANCE ASSESSMENTS.
12/11/13 • Entry Task • Describe the energy transfers and transformations from the gas coming through the tube to the paperclip in the flame. (Remember gas is stored chemical energy) • Homework: Read pages 200-210. Answer questions #1-10 on p. 211. Due Thursday 12/12/13
Today’s Agenda • Finish Performance Assessments and Bunsen Burner Licenses. • Once you receive your license….MAKE SURE I HAVE THAT RECORDED IN GRADEBOOK BEFORE YOU LEAVE! • Flame Test Lab – using colors to determine chemical elements. • NEVER LEAVE FLAME UNATTENDED. Make sure a team mate is attending the burner before you retrieve the chemical. • Test the chemical in the hottest part of the flame. • Put the paper clip back in the appropriate beaker!!!
General Steps for Flame Test • Make sure your flame test loop is clean. • Put it into hottest part of Bunsen Burner flame. No color change should occur if it is clean. • If it isn’t clean, dip it into acid provided, then put into flame. Repeat until there is no color change. • Dip clean loop into solution. • Put loop into hottest part of the Bunsen Burner flame. • Observe and record the color of the flame for the particular solution. • Put the paperclip back in SAME SOLUTION!
12/11/13 • Exit Task • CANCELLED DUE TO PERFORMANCE ASSESSMENTS. • IF TIME – What colors did the flame tests show? • Cobalt – • Magnesium – • Calcium – • Barium – • Potassium –
12/12/13 • Entry Task • Discuss yesterday’s flame test results. • Give a definition of DENSITY. • Get out homework. Calculate percent and QUICK review!
Classifying Matter Matter Are all the particles alike? YES NO Pure Substance Mixture Are the particles one kind of atom? Are the particles well-mixed and mixed evenly? YES NO YES NO Homogeneous Mixture Heterogeneous Mixture Compounds or Molecules Element
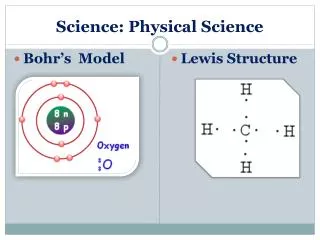
Pure Substances • Elements • all particles are alike • the smallest particle of an element that is still recognizable is called an atom • they can’t be broken down anymore by “normal means” • they are found on the periodic table • currently there are about 118 elements • chemical symbols are used to represent the elements • Example: C=carbon, N=nitrogen…..
Compounds and Molecules (over 10 million exist) • two or more elements chemically combined • once combined, the properties of a compound are different than the elements that make it up • can be broken down into the elements that make them up • example: Sugar is C6H12O6 • made up of carbon, hydrogen, and oxygen • looks nothing like C, H, O by themselves • can be broken down when burned
Mixtures • Heterogeneous Mixture • different samples are not necessarily made up of exactly the same proportions of matter • can often see different particles mixed together • often can be easily separated
Homogeneous Mixture • is the same throughout • often can’t see different particles mixed together • often difficult to separate • examples: steel, milk, salt water, Kool Aid®
properties of matter are that it has: • mass: amount of matter (atoms) in an object • volume: amount of space an object takes up • density: the mass per unit volume of an object
density is a property of material independent of quantity or shape
Density v D = m/v D = density m= mass v= volume
Example problem • A piece of lead has a mass of 56.4 g and a volume of 5 mL.Calculate its density. Density D = 56.4 g/ 5 mL D = 11.28 g/mL Same as… D = 11.28 g/cm3 Mass = 56.4 g Volume = 5 mL D = m/v ml is same as cm3 so sometimes see g/cm3