Aromatic Compounds
Aromatic Compounds. What is aromaticity. The Huckle Rule - compounds that contain (4n+2) pi electrons are considered to be aromatic (n is an integer) Anti-aromatic compounds contain 4n pi electrons Name comes from the strong aromas of these compounds. Benzene. Aromatic hydrocarbon C 6 H 6

Aromatic Compounds
E N D
Presentation Transcript
What is aromaticity • The Huckle Rule - compounds that contain (4n+2) pi electrons are considered to be aromatic (n is an integer) • Anti-aromatic compounds contain 4n pi electrons • Name comes from the strong aromas of these compounds
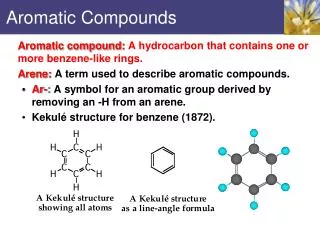
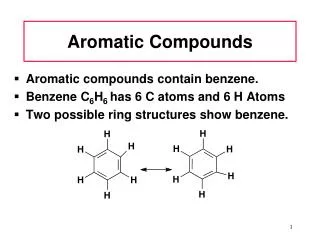
Benzene • Aromatic hydrocarbon • C6H6 • 6 pi electrons • Fully conjugated • Resonance stabilized
Structure of Benzene • All of the following are accepted representations of benzene
Molecular Orbitals of Benzene the benzene system has three molecular orbitals which accommodate the six electrons, two in each
Resonance Stabilization of Benzene • Cyclohexane – saturated • Benzene – unsaturated • Hydrogenation values • Cyclohexene 28.6 kcal/mole • Cyclohexadiene 55.4 kcal/mole • Cyclohexatriene 49.8 kcal/mole • Expected around 84-86 kcal/mole • Therefore: resonance stabilization energy ~30-36 kcal/mole
Experimental Evidence • Halogenation of benzene produces no reaction without a catalyst • Most substitution reactions of benzene are endothermic (absorb energy)
Monosubstitued Benzenes • Toluene • Bromobenzene • Chlorobenzene • Phenol • Nitrobenzene • Alkylbenzenes
Toluene • Toluene is a methylated benzene
Nitrobenzene and Alkyl benzenes Monosubstitued benzenes with a NO2 or alkyl group substituent
Benzene Activation vs. Deactivation • Activation - Once a substituent has been added to the benzene ring it can cause the molecule to be more reactive than the benzene molecule alone. • Deactivation – when a substituent on a benzene ring causes the molecule to be less reactive than benzene alone
Disubstituted Benzene When a second substituent adds to a benzene ring the designations are:
Ortho – Para DirectorsRing Activating • Alkyl groups • Halogens • OH • OCH3 • OR • NH2 • NHR • NR2
Meta DirectorsRing Deactivators • NO2 • HSO3 • COR • CN • COOH • COOR
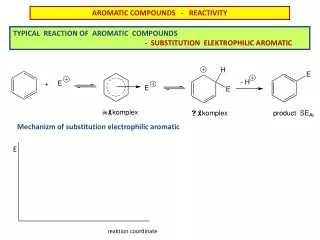
Reactions of Benzene • Electrophilic Aromatic Substitution - EAS is an organic reaction in which an atom, usually hydrogen, attached to an aromatic system is replaced by an electrophile. The most important reactions of this type that take place are nitration, halogenation, sulfonation, and acylation and alkylation.
Electrophile • an electrophile (literally electron-lover) is a reagent attracted to electrons that participates in a chemical reaction by accepting an electron pair in order to bond to a nucleophile • Most electrophiles are positively charged, have an atom which carries a partial positive charge, or have an atom which does not have an octet of electrons. .
Halogenation of Benzene Other catalyst AlCl3
Halogenation Mechanism Step 1
Halogenation (cont) Step 2 The hydrogen is removed by the AlCl4- ion which was formed in the first stage. The aluminium chloride catalyst is re-generated in this second stage.