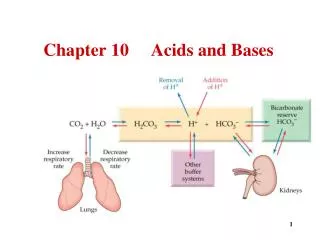
Intersection 10: Acids and Bases
Intersection 10: Acids and Bases. 11/7/06 Reading: 16.1 (p765-770) 16.3-16.7 (p773-794). Exam 1, Problem 12 Regrade. Please turn in your exams by noon on Tuesday, 11/14. A. Outline. Equilibrium wrap up From last week Two everyday examples Practice questions Acids and Bases History

Intersection 10: Acids and Bases
E N D
Presentation Transcript
Intersection 10: Acids and Bases 11/7/06 Reading: 16.1 (p765-770) 16.3-16.7 (p773-794)
Exam 1, Problem 12 Regrade Please turn in your exams by noon on Tuesday, 11/14
A Outline • Equilibrium wrap up • From last week • Two everyday examples • Practice questions • Acids and Bases • History • Models • pH Scale • Dissociation • Identifying acids and bases
A Equilibrium What are the characteristics of an equilibrium reaction? Use each of the following words in a sentence that describes an equilibrium reaction: products and reactants concentrations dynamic rates completion Keq Where does the “equal” in equilibrium come from?
A Can Equilibrium be Changed? Use LeChâtelier's Principleto predict what you will see: Fe(NO3)3 + KSCN ↔ Fe(SCN)+2 + KNO3DH < 0 red
M Q trumps LeChâtelier CaCO3(s) ↔ CaO(s) + CO2(g) ΔH > 0 Using each method, explain what will happen to the concentration of CO2 if solid lime (CaO) is added to the system?
M Equilibrium is Everywhere • Iodine thermometer Pictures from: jchemed.chem.wisc.edu/.../ 2003/Aug/abs878_1.html
A Equilibrium is Everywhere Salting the roads Is ice in equilibrium? http://antoine.frostburg.edu/chem/senese/101/solutions/faq/why-salt-melts-ice.shtml Picture from: www.glrc.org/story. php3?story_id=1377 What happens when salt is added to snowy winter roads?
A Question 1 Apatite, Ca5(PO4)3OH is the mineral in teeth. Ca5(PO4)3OH(s) 5 Ca+2(aq) + 3PO4-4(aq) + OH-(aq) On a chemical basis explain why drinking milk strengthens young children's teeth.
M Problem 1 Given the following equilibrium H2 + I2 2 HI Keq = 25 If you have 1 mol H2, 2 mol I2 and 3 mol HI in a 1 L flask, will you make more H2 or HI?
M Question 2 CrO4-2(aq) + 2H+(aq) ↔ Cr2O7-2(aq) + H2O(l) Explain what will happen to the equilibrium if water is added to this system?
A Acids and Bases What makes something acidic/basic? What do you know about acids and bases? Who can name the most?
A 2000 Top 20 Chemicals Produced in US* *It does not include minerals which do not require processing, such as salt and sulfur, and petrochemical feedstocks, such as ethane and butane, which are considered products of oil companies. http://scifun.chem.wisc.edu/chemweek/Sulf&top/Sulf&Top.html
A History of Acids and Alkalies www.catskillarchive.com/ dwellers/g.htm nefertiti.iwebland.com/ timelines/topics/drink.htm
A …a Brief History The only acid know to the ancient Egyptians, Greeks, and Romans was______? It was made by air oxidation of fermented fruit juice (wine) Among the alkalies known to the ancients were potash (potassium carbonate) obtained from____, soda (sodium carbonate) made by evaporation of alkaline waters, and lime (calcium oxide) made by roasting________. Caustic potash and caustic soda (potassium and sodium hydroxides) were made by the action of lime on soda and potash. Kauffman, G. B. "The Bronsted-Lowry Acid-Base Concept" J. Chem Ed. 1988, 65, 2831.
A …a Brief History Later, during the middle ages, the alchemists learned to make aqua frotis (nitric acid), aqua regia (a nitric-hydrochloric acid mixture), and oil of vitriol (sulfuric acid).
A …Glauber Mid-1600's Johann Rudolph Glauber 2 NaCl + H2SO4 2 HCl + Glauber’s salt (Na2SO4) Acid + base = salt + water KOH + HNO3 KNO3 + H2O “Liquor fixus (KOH or K2CO3 solution) and spiritus acidus nitri (HNO3) are in their nature…totally unlike, foes and adversaries of each other…and when the two are brought together…and the one part has overcome and killed the other, neither a fiery liquor nor a spiritus acidus can be found in their dead bodies, but the same has been made, as both were before and from which they were derived namely ordinary saltpeter (KNO3).”
M … All Acids and Alkali Theory Otto Tachenius and Francois Sylvius tried to simplify the chemistry of life processes by reducing all chemical interactions within the living organism to acid-base reactions. What evidence can you think of to support or discredit the theory of Tachenius and Sylvius?
M … Boyle Boyle (1663) noted that acids, in addition to their sour taste, had exceptional solvent power, the ability to color certain blue vegetable dyes red, and a precipitating action on dissolved sulfur. Alkalies, on the other hand, had a slippery feel and detergent properties, the ability to dissolve oils and sulfur, and the capacity to counteract acids and destroy their properties. Boyle's tests showed that some substances were neutral and did not classify either as acids or alkalies."
M … Lemery Nicholas Lemery (1675) described acids as having sharp spiky atoms, which produced a pricking sensation on the skin, and alkalies as being made up of round particles, which made them feel slippery or soapy. When acids and bases were mixed, he pictured the sharp needles of the acids as penetrating the porous alkali globules, thus producing salts, which were neither stinging nor slippery to the touch.
M Acids: Oxygen or Hydrogen? Antoine Lavoisier named the gaseous element oxygen in 1777. When sulfur or phosphorus was burned in oxygen, the products dissolved in water to form acids, so he concluded that oxygen was the element common to all acid materials. Claude Louis Berthollet (1789) showed that prussic acid (HCN) did not contain oxygen Humphry Davy proved Lavoisier's error more convincingly with muriatic acid (HCl), a very strong acid.
M Dualistic Theory Following the development of the battery by Alessandro Volta (1800), chemists began to use this new device to decompose all kinds of substances. Jons Jacob Berzelius and William Hisinger (1803) found that when salt solutions were subjected to electrolysis, bases were found at the negative pole and acids at the positive pole. They interpreted this to mean that acids and bases must carry opposite electrical charges. Berzelius concluded that acid-base reactions were simply the result of electrical attractions. His dualistic theory (1812) explained all chemical interactions in terms of neutralization of opposite electrical charges
M Arrhenius Model (PhD describing this work received lowest possible rating from his University) Svante August Arrhenius, during his study of electro-chemistry, observed that solutions of salts, acids, and bases were the only liquids that would conduct an electric current. He suggested (1884) that when these compounds dissolved in water they dissociated into charged particles, which he called "ions." According to the Arrhenius theory acids are compounds that produce hydrogen ions in water solution: HCl H+ + Cl- and bases are substances that provide hydroxide ions in water solution: NaOH Na+ + OH-
A H+ ions in water…Hydronium? HCl(aq) H+ (aq) (proton) + Cl-(aq) H2O H3O+ (aq)(hydronium)
A Hydronium H3O+ (H2O)6 ref 1 H3O+ (H2O)20 ref 2 H9O4+ref 3 H5O2+ref 4 Figures: http://itl.chem.ufl.edu/2045/lectures/lec_x.html http://cwx.prenhall.com/petrucci/medialib/media_portfolio/17.html • Zavitsas, A.A. (2001) Properties of water solutions of electrolyes and nonelectrolytes J. Phys. Chem. B105 7805-7815. • Hulthe, G.; Stenhagen, G.; Wennstrom, O.; Ottosson, C.H. (1997) Water cluster studied by electrospray mass spectroscopy. J. Chromatogr. A512 155-165. • Zundel, G.; Metzger, H. (1968) Energiebander der tunnelnden Ubershub-Protenon in flussigen Sauren. Eine IR-spektrokpische Untersuchung der Natur der Gruppierungen H5O2+Z. Phys. Chem. 58 225-245. • Wicke, E.; Eigen, M. Ackermann, Th. (1954) Uber den Zustand des Protons (Hydroniumions) in waBriger Losung Z. Phys. Chem. 1 340-364.
A Acid Base Reactions HCl (aq) + NaOH (aq) HNO3 (aq)+ KOH (aq) H2O (l) + NaCl (aq) H2O (l) + KNO3 (aq)
A Bronsted-Lowry Model Edward Franklin (1905) : NH4Cl + NaNH2 NaCl + 2 NH3 Thomas Martin Lowry in England and Johannes Nicholas Bronsted in Denmark (1923) independently arrived at a more inclusive definition of the neutralization reaction as the transfer of a hydrogen ion (a proton) from an acid to a base.
A Bronsted Lowry Practice HCl + NH3 HCl (aq) + NaOH (aq) HNO3 (aq)+ KOH (aq) H2O (l) + NaCl (aq) H2O (l) + KNO3 (aq) NH4Cl Acid: proton donor; Base: proton acceptor; Conjugate acid; conjugate base
A Acids and Bases: Comparing Definitions
+ If baseballs were really “base”balls….
M Measurement of Acids and Bases pH = -log[H3O+] HCl + H2O → H3O+ + Cl- A 1.0 M solution of HCl would produce 1.0 M H3O+ pH = -log [H3O+] = -log[ 1.0M] = 0
M How do you measure base [OH-]?
M Water undergoes an equilibrium process called autoionization.2 H2O(l) → H3O+(aq) + OH-(aq) • Write out the expression for the equilibrium constant (Kw) of this reaction. • In water, the [H3O+] and [OH-] ions are always in equilibrium with water having an equilibrium constant (Kw) of 1x10-14 • In pure water [H3O+] = [OH-] = 1x10-7 M. What about the pH?
M pOH pOH = - log[OH-] Remember that a low pH corresponds to a high concentration of H3O+ (acidic) solution. Therefore, a low pOH corresponds to a high concentration of OH- (basic) solution. Kw = [H3O+]*[OH-] = 1 x10-14 -log ([H3O+]*[OH-]) = - log (1 x10-14) -log [H3O+] - log[OH-] = 14 pH + pOH = 14
A Question 3: Can you explain the following?
A Problem 2 HA(aq) + H2O(l) → A-(aq) + H3O+(aq) What is the expression for the equilibrium constant? Ka = ([H3O+][A-]) [HA] Find the Ka if a 0.1 M HNO2 measures a pH = 2.2
A Vocabulary Strong (16 definitions): Weak (10 definitions): Favorable reaction.. ionizing freely in solution ionizing only slightly in solution Strong Reaction goes to completion spontaneous exothermic product favored
A Strong Acids Bases An acid that dissociates completely (the equilibrium is shifted all of the way to its conjugate base and hydronium ion) is said to be a strong acid. HCl(aq) + H2O(l) → H3O+(aq) + Cl-(aq) acid conj. base An acid that does not dissociate completely (an equilibrium is established in solution between the acid, its conjugate base, and hydronium ion) is said to be a weak acid. HClO2(aq) + H2O(l)↔ H3O+(aq) + ClO2-(aq) acid conj base Ka = ([H3O+][ClO2-]) / [HClO2]
A Strong Bases A base that dissociates completely (the equilibrium is shifted all of the way to its conjugate acid and hydroxide) is said to be a strong base. NaOH(aq) + H2O(l) → OH-(aq) + Na+(aq) baseconj. acid A base that does not dissociate completely (an equilibrium is established in solution between the base, its conjugate acid, and hydroxide) is said to be a weak base. (CH3)3N(aq) + H2O(l)↔ (CH3)3NH+(aq) + OH-(aq) baseconj. acid Kb = ([(CH3)3NH+][OH-]) / [(CH3)3N]
A Strong Acid Weak Acid
A There are six strongly dissociating acids: HCl HNO3 HBr HClO4 HI H2SO4 There are also five bases that dissociate completely in solution (strong): LiOH Ca(OH)2 NaOH Ba(OH)2 KOH You should commit the strong acids and bases to memory. Appendix F in your text book lists Ka and Kb values for many weakly dissociating acids and bases.
M Problem 3 Trimethylamine (CH3)3N has a Kb of 6.5 x10-5. Write out its chemical reaction with water: What is the [OH-] of a 0.010 M solution of triethylamine? What is the pOH? What is the pH?
M What kinds of hydrogen atoms (protons) are acidic? H-halogen (HF, HCl, HBr, HI) H2O H2S (Ka1 = 8.9x10-8) Oxoacids (H-polyatomic ions) (H2CO3, HNO3, etc.) HCN
M Organic Acids RCOOH
M Bases OH R3N
M Collecting Samples Obtain Whirl pack bags How do you fill the bags? How many samples should you take? How to store samples? Filter (acid wash all glassware) You may need special sampling techniques!
M Blanks • Field blank • Controls for contamination during travel • Lab Blank • Controls for contamination during analysis