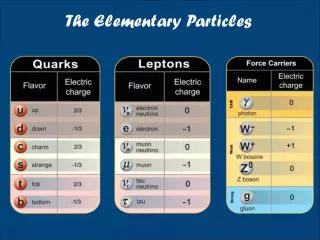
Elementary Particles
Elementary Particles. A Brief History to 1932 By Rick Dower Roxbury Latin School. Democritus (late 5 th Century B.C.). Supposed that the cosmos consisted of “atoms and the void” , i.e. very small indivisible particle and empty space. Aristotle (384 – 322 B.C.).

Elementary Particles
E N D
Presentation Transcript
Elementary Particles A Brief History to 1932 By Rick Dower Roxbury Latin School
Democritus(late 5th Century B.C.) • Supposed that the cosmos consisted of “atoms and the void” , i.e. very small indivisible particle and empty space.
Aristotle(384 – 322 B.C.) • Opposed atomism. Supposed the cosmos consisted of a plenum of infinitely divisible particles: Earth, Water, Air, Fire on Earth and Aether in the heavens.
Rene Descartes(1596 - 1650) • Followed Aristotle in believing the Cosmos is full of invisible particles. Supposed those particles whirling in vortices are responsible for gravity of Earth and planetary motion.
Isaac Newton(1642 - 1729) • Argued from the orbital motion of planets and comets that space contains no aetherial medium. “It seems probable to me that God in the Beginning form’d Matter in solid, massy, hard, impenetrable, moveable Particles” (Opticks, 1730).
John Dalton(1766 - 1844) • Proposed the idea of the “chemical atom” in New System of Chemical Philosophy (1808-1827) and gave first set of relative atomic weights based on combining proportions of elements in compounds.
Kinetic Theory of Gases • First proposed by Daniel Bernouli (1738). Rediscovered by John Herapath (1820) but rejected (opposed to Newton’s static model). Revived and refined by Rudolph Clausius (1856) James Clerk Maxwell (1859) and Ludwig Boltzmann. Atomic size and mass calculated from measurements.
Joseph John Thomson(1856 - 1940) • Experimentally determined the mass to charge ratio of cathode rays (electrons) and inferred that they are constituents of all chemical atoms. From an estimate of electron charge he determined an approximate mass of electron (about 1/2000 of H atom).
Pierre and Marie Curie(1859 - 1906) and (1867 – 1934) • Investigated radioactive elements, discovered polonium and radium, showed exponential decay of radioactive substance, measured atomic mass of radium, showed vast energy release of radioactive process.
Ernest Rutherford(1871 – 1937) • Named a and b radiation based on absorption of rays, explored radioactive decay series, proposed atomic transmutation in radioactive elements (1902), showed a particles are He nuclei, developed nuclear model of atom (1910) based on a particle scattering, demonstrated artificial transmutation (proton ejected when a collided with N nucleus, measured nuclear size.
Albert Einstein(1879 – 1955) • His 1905 paper on the photoelectric effect proposed that light energy comes in “quantum” units (later called photons). The energy of a photon is E = hf, where f = frequency of the light and h = Planck’s constant.
Photons • Robert Millikan’s experiments (1914-1916) verified Einstein’s photoelectric theory. • Arthur Holly Compton’s measurements of x-ray scattering by electrons in metals (1922-23) verified existence of photons.
James Chadwick(1891 – 1974) • In 1932 Chadwick showed that the penetrating radiation produced when beryllium was bombarded by fast a particles was an electrically neutral particle with mass about that of a proton, i.e. a neutron.
Neutrino • In 1914 Chadwick observed that electrons emitted in b decay have a continuous energy spectrum up to some maximum rather than a discreet energy. In 1930 Wolfgang Pauli proposed the emission of another particle (called a neutrino by Fermi) which shares energy with the b particle in the decay.
Cosmic Rays • In 1910 Father Theodor Wulf measured more radiation at the top of the Eiffel Tower than expected. In 1911-1912 Victor Hess made observations in balloons up to 5350 m above sea level. He showed that radiation increased with altitude. Robert Millikan verified Hess’s observations in the 1920s and called them cosmic rays.
Positrons • In 1932 Carl Anderson showed an observation of a cosmic ray particle in a cloud chamber that had characteristics of an electron but curved the opposite way in a magnetic field. He discovered the positrons predicted by Dirac’s theory of electrons.
Accelerators • John Cockcroft and Ernest Walton developed a voltage multiplying circuit to accelerate protons. In 1932 they disintegrated Li atoms into two a-particles as a result of bombardment by 770 kV protons. • Ernest Lawrence built his first successful cyclotron to provide acceleration in several small increments in 1930.