Mastering Ionic Chemistry: Formulas, Equations, and Stoichiometry Explained
Learn about ions, chemical formulas, equations, and stoichiometry in this comprehensive review. Understand oxidation numbers, monatomic and polyatomic ions, naming conventions for compounds, and predicting formulas of ionic compounds. Explore different types of molecules and chemical reactions. Master the fundamentals of advanced physical science and chemistry concepts.

Mastering Ionic Chemistry: Formulas, Equations, and Stoichiometry Explained
E N D
Presentation Transcript
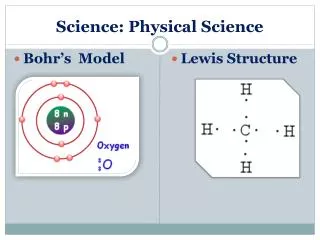
Advanced Physical Science B Chemistry in Review: The Ions Chemical Formulas Chemical Equations Stoichiometry
Ions • In general ions are formed from that lose or gain enough electrons to gain a full octet in their valence shell. • Elements that lose electrons become a positive CATION. • Elements that gain electrons become a negative ANION.
Monatomic Cations • Monatomic- one type of atom. • Most metals make monatomic cations, with a positive charge. • Usually the group number indicates the oxidation number of the elements in that group. • The cation simply has the same name as the element. • Transition Metals have multiple oxidation numbers.
Cu+1, Cu+2 Fe+2, Fe+3 Pb+2, Pb+4 Sn+2, Sn+4 Hg2+2, Hg+2 Cuprous, cupric Ferrous, ferric Plumbous, plumbic Stannous, stannic Mercurous, mercurric High -ic and Low -ous! Elements with Multiple Charge
Monatomic Anion • Monatomic- single type of atom. • Anions are usually made from Nonmetals, groups 15, 16, and 17. They gain electrons in their Valence. • All Anions end with a suffix. • Most monatomic anions end with a “-ide”.
Polyatomic Cations • Polyatomic- more than one atom. • There just a few polyatomic cations. • NH4+, ammonium • Hg2+2, mercury(I)
Polyatomic Anions • Polyatomic anions have more than one atom. • A nonmetal plus oxygen or oxygen and hydrogen. • Sometimes called an “oxyanion.” • Anions end with a suffix. • Most end with “-ate” • Polyatomic anions with less oxygens end with “-ite” • “ite” anions usually have one less oxygen then “ate” anions. • “ate” ate the “ite”!
Chemical Formulas The building blocks.
Symbols and Formulas • Names of Elements - 109 elements, >10 million known compounds • Compounds are represented by formulas combining chemical symbols and numeric subscripts. • Some elements are named for their properties. • Nitrogen-“niter forming” • Plumbic (lead)-shiny • Some elements are named for their place of origin.
Symbols and Formulas (cont.2) • Some elements are named for the minerals they are found in. • Tungsten-Swedish name for “heavy stone” • Some elements are named in honor of a person. • Symbols for the elements • One or two letters, the first letter is capitalized • In 1813, JJ Berzelius, a Swedish chemist developed the modern symbols for designated elements.
Chemical Formulas • Are a combination of symbols that represent the composition of a compound. • Molecular Compounds and Ionic Compounds.
Ionic Compounds • Are compounds composed of charged particles. • In general: the electrons are shared between the ions. Metals tend to give up their electrons to an incomplete nonmetal. • All Ionic compounds are represented by their empirical formulas. They are always in the smallest whole number ratios.
Other Types of Molecules • Diatomic Molecules:these 7 elements must exist in nature paired with itself or other elements. • Br2, Bromine - O2, Oxygen • I2, Iodine - H2, Hydrogen • Cl2, Chlorine - N2, Nitrogen • F2, Fluorine • “BrIClFOHN”
Other Types of Molecules (cont.2) • Hydrates:Ionic Molecules attached to water molecules. • Organic Molecules:contains carbon as it’s central element. • Alloys: metals form these molecules where atoms are held together by a “sea” of electrons.
Predicting Formulas of Ionic Compounds • Write the symbols for the elements in the compound • Always write the CATION first. • Determine the charge on each ion. • Na+1=+1, O-2=-2 • From the charge on each ion use subscripts to indicate the multiplier for the ions. • The total positive must equal the total negative. • The “total” charge of the compound must be zero. • Ex. Na2O
Predicting Formulas of Ionic Compounds (cont.2) • When using subscripts for polyatomic ions, the ion is placed in parentheses, and the subscript is placed on the outside to indicate “x” ion units. • The subscript applies to all the elements in the parentheses. • If the subscript is “1”, it is understood and not written. • For monatomic ions no parentheses is used.
Naming Ionic Compounds • Naming Binary Ionic Compounds: • The cation is listed first, then the monatomic anion. • For stock names include the oxidation number of the cation in parantheses. • For traditional names use the “-ous” or “-ic” name for the cation.
Naming Ionic Compounds (cont.2) • Naming Ternary Ionic Compounds: • Made up with a cation and a polyatomic anion. • The suffix tells which anion. • “-ate” for more oxygen's • “-ite” for less oxygen's.
Chemical Equations A chemical recipe
Types of Chemical Reactions • There are 5 fundamental types of Chemical Reactions. • Synthesis (Direct Combination) • Decomposition (Analysis) • Single Replacement • Double Replacement • Combustion
Synthesis(Direct Combination) • “joining together” • The general form of reaction: • A + B AB • element+element compound • Two reactants One product
Synthesis (cont.2) O2 + 2NO2NO2
Decomposition (Analysis) • “breaking down” • The general form of reaction: • AB A + B • compound element + element • One reactant Two products
Decomposition (cont.2) 2NI3 N2 + 3I2
Single Replacement • “Like ions must displace like ions” • The general form of reaction: • A + BC AC + B • element + compound compound + element • Two reactants Two products
Single Replacement (cont.2) Fe2O3 + 2Al Al2O3 + 2Fe
Double Replacement • “Exchanging ions” • The general form of reaction: • AC + BD AD + BC • compound+compound compound+compound • Two reactants Two products
Double Replacement (cont.2) AgNO3 + NaCl AgCl + NaNO3
Combustion • Special form of a decomposition rxn. • Burning hydrocarbons. • Metabolism • The general form of reaction: • hydrocarbon + oxygen CO2 + H2O • Presence of oxygen in the form, O2 • Products are always CO2 and H2O
Combustion (cont.2) 2C8H18 + 17O2 18H2O + 8CO2
Special Considerations for Replacement Reactions • Single Replacement Reactions: follow the “Activity Series” of elements. • Cations displace cations. • Anions displace anions. • Li+1 is the most reactive cation. • F-1 is the most reactive anion. • Double Replacement Reactions: must show evidence of a chemical reaction. • “God Punishes Chemistry Teachers” • Gas, Precipitate, Color change, Temperature change.
Atom Accounting • Reactants-a starting substance in a chemical reaction. • Products-a substance produced in a chemical reaction. • Atoms in the reactants must equal the atoms in the products.
Balancing Chemical Equations • Do an “Atom Accounting” • H2 + N2 NH3 H=2 H=3 N=2 N=1 • Li + Al2(SO4)3 Li2SO4 + Al Li=1 Li=2 Al=2 Al=1 S=3 S=1 O=12 O=4
Balancing Basics • Rules for Balancing Chemical Equations: • Law of Conservations of Matter: “What goes IN must come OUT” • Be sure the elements in the products are in the reactants. • Make sure COMPOUNDS are good chemical formulas. • Use subscripts to make formulas.
Balancing Basics (cont.2) • Balance the atoms on each side of the equation using COEFFICIENTS. • Do NOT Touch the Subscripts! • Keep the coefficients in the lowest whole numbered ratios. • Ex. 4H2 + 2O2 2H2O • Will be: • 2H2 + O2 H2O
Balancing Basics (cont.3) • Balance the equation: • 3H2 + N22NH3 H=2 6 H=3 6 N=2 N=1 2
Balancing Basics (cont.4) • Li + Al2(SO4)3 Li2SO4 + Al Li=1 Li=2 Al=2 Al=1 S=3 S=1 O=12 O=4 • 6Li + Al2(SO4)33Li2SO4 + 2Al Li=1 6 Li=2 6 Al=2 Al=1 2 S=3 S=1 3 O=12 O=4 12
Stoichiometry Mathematical with chemical equations.
Stoichiometry • A Chemical Equation gives information about the relative relationship (ratio) between reactants and products in a chemical reaction. • Coefficients of a balanced chemical equation gives three pieces of quantitative information about the reactants and the products. • The relative number of particles. • The relative number of moles. • The relative volume of a gas, at the same temperature and pressure.
Stoichiometry (cont.2) • When a chemical equation is balanced, the total mass of the reactants equals the total mass of the products. (Law of Conservation of Mass) • The coefficients DO NOT give relative ratios of reactants to products by mass. • Must convert to MOLE or particles the compare coefficients.
Stoichiometry (cont.3) • Organization is critical. • Balance the chemical equation, FIRST! • Determine the element/compounds that is given and the element/compound that is sought. Make a chart. • Place the information given in the problem under the correct element/compound.
Mixed Stoichiometric Relationship • In general, this relationship holds: • All mixed relationship problems take 3 steps. • First, always balance the chemical equation and organize the problem. Determine what is Given and what if Sought. • Convert Given to moles, Change Given to Sought, Convert from Sought moles to whatever units asked for. • Remember: • If changing to/from mass: 1mol=Molar Mass • If changing to/from particles: 1mol=6.02x1023parts • If changing to/from volume(gases only): 1mol=22.4dm3 at STP
Mixed Stoichiometric Relationship (cont.2) • MOLES RULE!!! • In One DA Table: Xunit,Sought = Given,units CF1 CF2 CF3 • Where: • CF1=converts units Given to moles • CF2=converts moles Given to moles Sought. The Mole Bridge. • CF3=converts moles Sought to units Sought.
Try this mass-mass problem: • Calculate the mass of oxygen produced if 2.50g of potassium chlorate is completely decomposed to give potassium chloride and oxygen. • Balance the Chemical Equation: • 2KClO3 2KCl + 3O2 • Determine the Given and the Sought: • Given: 2.50g KClO3 • Sought: mass of O2 produced • Organize the appropriate information: • 2.50g KClO3 Xg O2
In One Step • Calculate the mass of oxygen produced if 2.50g of potassium chlorate is completely decomposed to give potassium chloride and oxygen.. • The balanced chemical equation: • 2KClO3 2KCl + 3O2 • XgSought =Givenmass 1molGiven Mole MolarMassSought MolarMassGiven Bridge1mol Sought • XgO2 = 2.50gKClO3 1molKClO3 3O2 32gO2 123gKClO3 2KClO3 1molO2 = 9.76x10-1gO2
#2 Try this: • How much silver phosphate is produced if 10.0g of silver acetate reacts with sodium phosphate? • Balance the Chemical Equation. • Organize the problem. • Use Three or One Step to solve the problem.
How much silver phosphate is produced if 10.0g of silver acetate reacts with sodium phosphate? 3AgC2H3O2+ Na3PO4 Ag3PO4+ 3 NaC2H3O2 10.0gAgC2H3O2 XgAg3PO4 XgAg3PO4=10.0gAgAce 1AgAce 1Ag3PO4 418.58gAg3PO4 166.92gAgAce 3AgAce 1Ag3PO4 X= 8.36gAg3PO4