B. Water
B. Water. Needed by but not c reated by living things. Created by living things Contain carbon as central atom Also contain hydrogen. Water – inorganic molecule, essential to ALL life. 1 . Chemistry of Water

B. Water
E N D
Presentation Transcript
B. Water • Needed by but not • created by living things • Created by living things • Contain carbon as central atom • Also contain hydrogen
Water – inorganic molecule, essential to ALL life 1. Chemistry of Water • Water is bonded by a polar covalent bond = electrons are unequally shared between O and H • OVERALL – the molecule has a polar nature • Meaning – one end is slightly positive and one end is negative
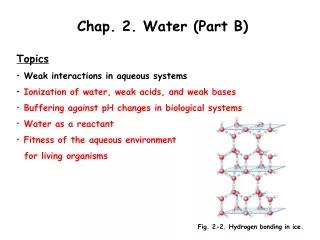
2. Hydrogen Bonding • Between water molecules there are HYDROGEN bonds • Definition – H –Bonds A chemical bond in which a hydrogen atom of one molecule is attracted to an negative atom, - of another molecule
A. Properties of Water Adhesiveness • Creates surface tension- aquatic insects can travel on surface High Specific Heat Capacity • help regulate body temperature • Water requires a large amount of energy to changes temperature
Properties (cont.) Universal solvent – water will dissolve many solutes -blood carries dissolved solutes -oceans carry dissolved solutes Transparent in Nature- Aquatic environments for photosynthesis Lubricant – swallowing, joints
II. Acids, Bases and Buffers When water DISSOCIATES H = OH This is a neutral solution (pH 7)
Acids • When the concentration of the H ion is greater than the concentration of the OH ion then we have an acid. • Ex. HCl in stomach • Ex. Lemon juice • How do acids taste??
Bases • When the [ OH] is greater than [H] then we have a base. • Ex – ammonia, oven cleaner
pH & pH Scale • is a measure of the concentration of hydrogen ions that are free in a system • High [H] LESS pH • LowRanges from 0 to 14 pH Scale • 0 to 7 = Acidic • 7 = neutral = water • 8-14=basic or alkaline
Buffers • A compound that tends to maintain a solution at a constant pH by accepting or releasing hydrogen ions • Ex. Bicarbonate ion