The Ozone Layer Chapter 15
The Ozone Layer Chapter 15. The ozone layer = 15-30 km altitude in stratosphere Photochemical smog is ozone in the wrong place Earth’s Natural Sunscreen: blocks ultraviolet light Ozone is only a small fraction of the gases N 2 , O 2 are still the majority of the gases in ozone layer

The Ozone Layer Chapter 15
E N D
Presentation Transcript
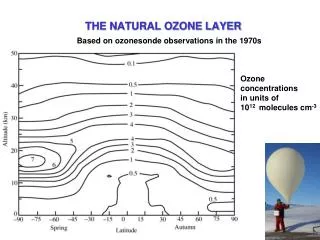
The ozone layer = 15-30 km altitude in stratosphere Photochemical smog is ozone in the wrong place Earth’s Natural Sunscreen: blocks ultraviolet light Ozone is only a small fraction of the gases N2, O2 are still the majority of the gases in ozone layer O3 would make a 3.5 mm thick layer at Earth’s surface Ozone is produced at the tropics, flows to poles Good Ozone
Antarctic Ozone Hole • Ozone totals are measured in “Dobson Units” • 350 Dobson Units is typical in temperate regions • Ozone levels have decreased below 150 Dobson Units over Antarctica • Began to occur in late 1970’s • Levels are lowest in Antarctica’s Spring (Sept—Nov) • Normal levels regained by June—July • Geographic Region effected is growing each year • Not really a hole; more like a thinner window
Cause of the Ozone Hole • Chlorine Pollution has been proven as the cause • CFC’s = Chlorofluorocarbons • One of the main sources of atmospheric chlorine • Used as refrigerants in air conditioners and refrigerators • Fairly unreactive, so they make it to ozone layer • Laws have greatly reduced chlorine pollutions • We will probably still have ozone hole until ~2050
Molecules and Light • The spectrum • Visible light: 400—750 nm (ROYGBIV = rainbow) • Color changes as wavelength changes • Ultraviolet light = UV = 50—400 nm (invisible) • More energetic than visible light • Energy increases as wavelength decreases • Molecules can interact with visible and UV light • Absorption Spectrum = graph of light absorbed vs wavelength • Light absorption increases the energy of the molecule • Bonds can be broken or new bonds formed • O2, as all molecules do, absorbs light selectively
A portion of the electromagnetic spectrum O2 absorption spectrum in UV
Ozone absorbs UV light • O2 absorbs all UV between 120—220 nm • Above the stratosphere • UV between 50—120 nm is absorbed by N2, O2 • O3 vital because it absorbs 220—320 nm UV • UV-C = 220—280 nm O3 + O2 remove all • UV-B = 280—320 nm O3 absorbs 70-90% • Depends on season, latitude, etc… • Never completely successful • Fraction of UV-B escaping increases as wavelength increases • UV-A = 320—400 nm is not stopped by any gases • Least harmful type of UV light
Biology and the Ozone Layer • UV-B does escape the ozone layer • 1% loss in ozone = 2% increase in UV-B on the ground • Causes sunburn, cataracts, and skin cancer • Affects immune system • Affects the growth of plants and animals • DNA absorbs UV-B and is damaged • Replication of new DNA, cells goes wrong = cancer • Skin cancer is most common, since it gets most sun • Loss of ozone is predicted to cause increase in skin cancer • Slow spreading form; can be detected and treated • 25% of Americans will have some form in their lifetimes
Malignant Melanoma • Fatal form of skin cancer • 1 in 100 Americans will likely get it • Short, high UV exposure early in life linked to it • Fair skin, fair hair, freckles makes you susceptible • 15-25 year lag time between exposure and melanoma • Likely only seen the tip of the iceberg • Skin cancer cases are expected to rise (1970’s + 25 = 2000) • Incidence of melanoma based on several things • Latitude: Texas and Florida have higher rates • Lifestyle: Australia has higher rates
Sunscreens • Substances that block UV light on your skin • Some only block UV-B, not UV-A • Don’t get burned, but can still damage your skin, DNA • How they work • Reflect all light = ZnO, TiO2; white, inorganic • Absorb UV before it reaches your skin = organic molecules • Must not break down as they absorb light ----> useless Octyl methoxycinnamate (OMC) 4-Methylbenzylidene camphor (4-MBC)
Other Effects of UV • Cataracts • Cornea and Lens filter 99% of UV before it reaches your retina • Chemical reactions cause opaqueness of the lens • Primarily a UV-B caused problem • Usually long-tern; short-term when severe exposure • Effects on Animals and Plants • Photosynthesis by plants reduced when UV-B increases • Oceans depend on surface phytoplankton • Amphibian mortality and deformity
Normal Eye Eye with a cataract
Ozone Hole Chemistry • Light Energy • Photon = discrete packet of light having a specific energy • Energy of light is inversely related to the wavelength • Infrared = wavelength longer than visible is less energetic • Ultraviolet = wavelength shorter than visible is more energetic • Energy: UV-C > UV-B > UV- A > Visible > Infrared • Photochemical Reaction • Requires light energy to start a chemical reaction • Light must be of exact wavelength/energy required • 500 nm light activated reaction won’t work with 400 nm
Creation of Ozone • Ozone formation occurs in the upper stratosphere • UV-C not yet filtered out at this altitude O2 + UV-C ----> 2 O O + O2 ----> O3 + heat • Temperature of stratosphere is warmer than those below • Temperature Inversion (usually cools as you move upward) • Little mixing of the O3 layer that forms
Recycling of Ozone • Ozone is destroyed as it filters UV O3 + UV-B/UV-A ----> O2 + O O2 + O ----> O3 (mostly) O3 + O ----> 2 O2 (some) • NO and other gaseous pollutants speed up O3 destruction • O3 “lives” about 30 minutes at 30 km altitude • Formation/Destruction is called the Chapman Cycle • Ozone Layer: < 10 ppm O3 << O2 and N2
Chlorine and Ozone • Chlorine initiates ozone destruction Cl + O3 ----> ClO + O2 (chorine monoxide) 2 ClO ----> ClOOCl (dichloroperoxide) ClOOCl ----> ----> ----> 2 Cl + O2 Total: 2 O3 ----> 3 O2 • Cl is a catalyst (speeds up reaction); not used up • Each Cl atom can destroy 50 O3 molecules per day • Bromine (Br) reacts in the same way • Small amount compared to Cl pollution
Chlorine Activation • Only Cl, ClO are active ozone destroying Cl forms • Cl is mostly found in inactive form • ClONO2 = chlorine nitrate ClO + NO2 ----> ClONO2 • HCl = hydrogen chloride Cl + CH4 ----> HCl + CH3 • Antarctica’s weather favors active forms • Dark/cold: PCS = polar stratospheric clouds (ice crystals) • Air pressure drops = vortex = whirling cold air is isolated • HCl + ClONO2 + PCS ----> Cl2 + HNO3 (dark) • Cl2 + light ----> 2 Cl (beginning of spring) • Ozone decreases 2% per day until PCS clouds melt
An Arctic Ozone Hole? • Arctic winters are not as cold as Antarctic ones • Vortex breaks up before light is present to activate Cl • Temperatures are expected to drop in Arctic • 1996 record cold year • Observable “hole” in Arctic Ozone occurred • Chlorine pollution is dropping worldwide • Enough in atmosphere for Arctic Hole for 10-20 years
Ozone Decreases in Non-Polar Areas • Worldwide ozone amounts fell about 3% since 1980 • Besides poles, mid-latitudes experienced greatest loss • Decreases usually occur in March—April in N. Hem. • The explanation of this phenomenon is not as clear • Do pollution droplets function like polar PCS clouds? • Volcanoes have given off H2SO4 in these regions • Dilution of Polar air masses definitely contributes
Ozone Depleting Chemicals • Sink = natural process to regulate the concentration of a compound: CO2 has vegetation, oceans as a sink • Sources of Chlorine and Bromine • CH3Cl (methyl chloride) produced by decaying plants • CCl4 (carbon tetrachloride) used in dry cleaning • Manmade CFC’s have no sink in nature • Not water soluble • Doesn’t react with most other gases • Not reactive with visible or UV-A light • Drift to stratosphere by natural buoyancy (60 yr lifetime) • CF2Cl2 + UV-C ----> CF2Cl + Cl
CFC Replacements • The presence of C—H bonds allows decomposition OH + H—CCl3 ----> H2O + decomposed products • CH3Cl is partially removed before reaching stratosphere • HCFC’s = hydrofluorochlorocarbons • CHF2Cl = HCFC-22 • Air conditioners and Refrigerators currently use these • Only about 5% of the risk to ozone as CFC’s • Temporary bridges to even better compounds • HFC’s = hydrofluorocarbons • FCH2CF3 = HFC-134a already being used
Ozone Hole: the success story • International Agreements have limited damage • Rowland/Molina predicted problem in 1970’s (Nobel) • CFC’s banned in Europe by late 1970’s • 1987 Montreal Protocol: phase out of ozone depleters • 1995 all legal CFC production ceased • 2010 goal for developing countries • Halon = CF3Br, CF2BrCl illegal by 1994 • HCFC use to end by 2030—2040 (no increase after 2015) • Future: chlorine pollution will be removed as HCl • Cl peak 1999 • No ozone hole after 2050